Levitra enthält Vardenafil, das eine kürzere Wirkdauer als Tadalafil hat, dafür aber schnell einsetzt. Männer, die diskret bestellen möchten, suchen häufig nach levitra kaufen ohne rezept. Dabei spielt die rechtliche Lage in der Schweiz eine wichtige Rolle.
Grna-61-11-06 1166.1170
Journal of Gerontology: MEDICAL SCIENCES
Copyright 2006 by The Gerontological Society of America
2006, Vol. 61A, No. 11, 1166–1170
Exercise: An Active Route to Healthy Aging
Aerobic Exercise Training Increases
Brain Volume in Aging Humans
Stanley J. Colcombe,1 Kirk I. Erickson,1 Paige E. Scalf,1 Jenny S. Kim,1 Ruchika Prakash,1
Edward McAuley,2 Steriani Elavsky,2 David X. Marquez,2 Liang Hu,2 and Arthur F. Kramer1
1Beckman Institute & Department of Psychology and 2Department of Kinesiology,
University of Illinois, Urbana.
Background. The present study examined whether aerobic fitness training of older humans can increase brain volume
in regions associated with age-related decline in both brain structure and cognition.
Methods. Fifty-nine healthy but sedentary community-dwelling volunteers, aged 60–79 years, participated in the 6-
month randomized clinical trial. Half of the older adults served in the aerobic training group, the other half of the olderadults participated in the toning and stretching control group. Twenty young adults served as controls for the magneticresonance imaging (MRI), and did not participate in the exercise intervention. High spatial resolution estimates of grayand white matter volume, derived from 3D spoiled gradient recalled acquisition MRI images, were collected before andafter the 6-month fitness intervention. Estimates of maximal oxygen uptake (VO2) were also obtained.
Results. Significant increases in brain volume, in both gray and white matter regions, were found as a function of
fitness training for the older adults who participated in the aerobic fitness training but not for the older adults whoparticipated in the stretching and toning (nonaerobic) control group. As predicted, no significant changes in either gray orwhite matter volume were detected for our younger participants.
Conclusions. These results suggest that cardiovascular fitness is associated with the sparing of brain tissue in aging
humans. Furthermore, these results suggest a strong biological basis for the role of aerobic fitness in maintaining andenhancing central nervous system health and cognitive functioning in older adults.
BEGINNING in the third decade of life the human brain among others (12). The end result of these structural
shows structural decline, which is disproportionately
changes is a better interconnected brain that is more plastic
large in the frontal, parietal, and temporal lobes of the brain
and adaptive to change (8,13). Given that cardiovascular
(1). This decline is contemporaneously associated with
exercise has similar effects on human cognitive function that
deterioration in a broad array of cognitive processes (2).
might be predicted from the structural changes in nonhuman
Given the projected increase in the number of adults sur-
animals, it seems likely that similar structural changes
viving to advanced age, and the staggering costs of caring
would be engendered in human brain tissue following
for older individuals who suffer from neurological decline,
chronic exercise, but research examining the impact of
identifying mechanisms to offset or reverse these declines
exercise on brain structure has overwhelmingly relied upon
has become increasingly important.
nonhuman animals, due to the highly invasive methods
Cardiovascular exercise has been associated with im-
typically required to assess changes in brain structure.
proved cognitive functioning in aging humans (3,4). These
With the advent of noninvasive in vivo brain imaging
effects have been shown to be the greatest in higher order
technologies such as structural and functional magnetic
cognitive processes, such as working memory, switching
resonance imaging (MRI), it is possible to address questions
between tasks, and inhibiting irrelevant information, all of
about changes in the underlying brain structure of humans.
which are thought to be subserved, in part, by the frontal
In one such study (14), we found that older adults with a
lobes of the brain (3). However, very little is known about
lifelong history of cardiovascular exercise had better pre-
the structural brain changes, if any, which underlie these
served brains than did age-matched sedentary counterparts.
benefits in humans. Previous research with nonhuman
Interestingly, the structural preservation was greatest in the
animals has shown that chronic aerobic exercise can lead
frontal and parietal regions of the brain, which are thought
to the growth of new capillaries in the brain (5,6), increase
to subserve aspects of higher order cognition, such as working
the length and number of the dendritic interconnections
memory, task switching, and the inhibition of irrelevant
between neurons (7), and even increase cell production in
information. However, owing to the cross-sectional nature
the hippocampus (8). These effects likely result from
of that study, it is conceivable that a number of factors
increases in growth factors such as brain-derived neuro-
influence both brain volume and aerobic fitness. It is even
trophic factor (7,9) and insulin-like growth factor (10,11),
possible that the relationship is reversed. That is, those older
FITNESS AND BRAIN HEALTH
Table 1. Demographic Information on Aerobic Exercising and
participate in either an aerobic exercise program or a non-
Nonaerobic Exercising Control Older Adults
aerobic stretching and toning exercise program.
Participant characteristics are documented in Table 1. The
Measured Variable
only significant difference between the aerobic and non-
aerobic training group participants was in the maximal oxy-
t(58) ¼ 2.05, p , .025
2) change measure (i.e., the cardiovascular
improvement from pre- to post-training). The Institutional
Review Board at the University of Illinois approved this
research. Written informed consent was obtained from all
Notes: All values except the change in VO2 outcome represent participant
Exercise Intervention Protocols
characteristics at the onset of study participation.
The aerobic exercise intervention was designed to
HRT includes participant's self-report of either opposed or unopposed
improve cardiorespiratory fitness with an exercise intensity
estrogen therapy, and participants included in the hypertensive category were
prescription derived from peak heart rate (HR) responses to
those who were diagnosed as hypertensive prior to their participation in thestudy.
baseline graded exercise testing. Intensity levels began at
Standard errors are in parentheses.
40%–50% HR reserve increasing (15) to 60%–70% HR
MMSE ¼ Mini-Mental State Examination score; NS ¼ not significant;
reserve over the course of the trial. Intensity levels and
HRT ¼ hormone replacement therapy.
exertion were recorded in daily exercise logs and monitoredby trained exercise leaders. Participants in the older
adults who have relatively well preserved brains may be
nonaerobic exercise control group followed the same
differentially able to maintain participation in a physically
activity schedule and format as the aerobic exercise group
active lifestyle, through better preserved cognitive abilities
did, but engaged in a program of whole-body stretching and
or some other set of genetic or environmental variables that
toning designed for individuals 60 years old or older. As the
affect both somatic and brain health.
individual's level of flexibility increased, stretches with
To address this issue, we randomly assigned 59 older
increasing levels of difficulty were incorporated into the
adults to participate in either a cardiovascular exercise group
program. Participants in both the aerobic and control
or a nonaerobic exercise control group for a 6-month period.
exercise groups attended three 1-hour exercise training
We scanned these participants in a high-resolution structural
sessions per week for the 6-month period of the inter-
MRI protocol immediately before and after participation
vention. Compliance in the exercise sessions was excellent,
in the exercise program. We then compared changes in
exceeding 85% for all participants. Each group participated
regional brain volume from preintervention to postinterven-
in their sessions at separate geographical locations around
tion for aerobic exercisers and nonaerobic exercise control
campus to reduce the probability of any crossover effects
participants using an optimized voxel-based morphometric
occurring between the groups.
technique which can assess tissue volume in a point-by-point fashion throughout the brain (see Methods). We
Assessment of Cardiorespiratory Fitness
additionally analyzed high-resolution brain scans of 20
Participants completed a graded exercise test on a motor-
younger adults; these scans were collected at the same
driven treadmill. Peak oxygen uptake (VO2peak) was mea-
intervals as those from the older adults. The younger adults
sured from expired air samples taken at 30-second intervals
did not participate in an exercise intervention, and served
until the highest VO2peak was attained at the point of voli-
largely as methodological controls as we did not expect to
tional exhaustion. The aerobic fitness training group showed
see any appreciable change in the volume of younger adult
a significant 16.1% in increase in VO2peak, whereas the older
brains within the 6-month time frame of the study.
control participants showed a nonsignificant 5.3% change inVO2peak across the 6-month intervention.
Imaging Protocols and Analyses
We acquired a high-resolution T1 weighted structural
image for each participant, 1 week prior to the intervention
Fifty-nine older (60–79 years) and 20 younger (18–30
and within 1 week after cessation of the exercise program.
years) right-handed, neurologically intact adults took part in
Twenty-two of the older adults and eight of the younger
the 6-month study. All participants were screened for
adults were scanned in a 1.5 Tesla GE Signa MRI scanner
neurological defect (e.g., possible dementia, self-report of
(1 3 1 3 1.3 mm; Niskayuna, NY) at both times 1 and 2 and
neurological disease such as multiple sclerosis, brain tumor,
the remaining older and younger adults were scanned in a
and Parkinson's disease) and appropriateness for testing in
3 Tesla Siemens Allegra MRI scanner (1 3 1 3 1.3 mm;
an MRI environment (e.g., no metallic implants that could
Malvern, PA) at both times 1 and 2. None of the results
interfere with testing, no claustrophobia). Older adults were
reported in this study were significantly impacted by the
additionally required to obtain physician approval for
scanner type used to acquire the MRI images.
participation in an exercise program before beginning any
Our voxel-based morphometry analyses largely followed
phase of the study. Older participants were randomly as-
those methods described elsewhere (16), with the exception
signed by the project coordinator during recruitment to
that we adapted our protocol to include a highly optimized
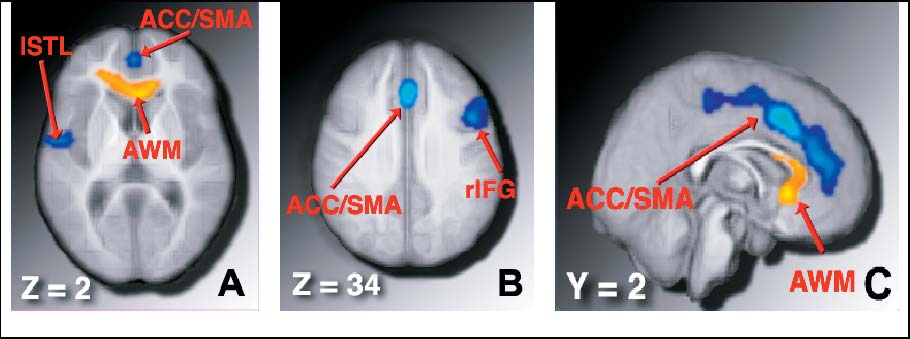
COLCOMBE ET AL.
Figure 1. Regions showing a significant increase in volume for older adults who participated in an aerobic fitness training program, compared to nonaerobic
(stretching and toning) control older adults. A and B, Neurologically oriented axial slices through the brain, at þ2 and þ34 mm, respectively, in stereotaxic space. C,Sagittal slice 2 mm to the right of the midline of the brain. Blue regions: Gray matter volume was increased for aerobic exercisers, relative to nonaerobic controls.
Yellow regions: White matter volume was increased for aerobic exercisers, relative to controls. (See also Table 2.)
and robust longitudinal registration approach to perform the
a set of unpaired t tests at each voxel. We initially subjected
initial coregistration between participants' time 1 and time 2
the younger adult data to a simple t test against zero to
images (17). The registration constrained spatial scaling by
evaluate whether any changes occurred during the 6-month
the skull to minimize any potential differences in scanner
period for younger adults. These analyses yielded three
geometry or misregistration due to soft-tissue changes.
statistical parametric maps for gray and white mater, which
First, each participant's images were skull-stripped and
described where (a) aerobic exercisers showed a greater
segmented into 3D maps of gray matter, white matter, and
increase in volume than stretching and toning controls, (b)
cerebrospinal fluid, using a semi-automated algorithm that
nonaerobic controls showed a greater increase in volume
takes into account voxel intensity distributions as well as
than aerobic exercisers, and (c) any change in volume,
hidden Markov random fields to estimate tissue volume at
positive or negative, was present in younger adults. We
each voxel (18). Then, the 3D maps of gray and white
performed a second set of analyses to examine whether the
matters for each participant were registered to a common
results of our initial analysis interacted with the two
space (MNI) using a 12-parameter affine transformation.
different MRI scanners used in the study. In none of the
These segmented images were then used as a priori
regions presented in Figure 1 did the scanner used to collect
templates for a second-level segmentation. In addition,
the MRI data interact with the effects of interest. The
a mean image was calculated from all participants, spatially
resulting statistical parametric maps presented in Table 2
smoothed with a 12 mm full-width at half max kernel, and
were statistically corrected for multiple comparisons at a
subsequently used as a study-specific template. The use of
p , .05 level for each cluster (19).
study-specific templates has been shown to reduce errorassociated with misregistration and, therefore, to provide a
better estimate of brain volume differences between groups.
Descriptive information on the participants is presented
The second-level analysis then consisted of a resegmentation
in Table 1. Participant ages ranged from 60 to 79 years, with
based on the a priori gray and white matter maps from stage
a mean of 66.5 years. Overall, the sample was 55% female,
1 and a realignment to the study-specific template image.
and tended to be well educated, with an average 13.8 years
These images provide a voxel-by-voxel estimation of the
of education. The estimated VO
volume of gray matter, white matter, and cerebrospinal fluid
2 scores ranged from 12.6 to
contained within the particular voxel. These images werethen multiplied by the Jacobian determinant for each
Table 2. Cluster Size, Peak Location, and Statistical Value for
participant to preserve original volume and to control for
Each of the Four Regions Where Aerobically Exercising
differences in the extent of registration and possible
Older Adults Showed a Significant Increase in Brain Volume
interpolation error. Finally, the percent change in volume
was computed at each voxel for each participant. All of
these processes were conducted by an experimenter who
was blind to the group assignment of each individual.
The maps representing the percent volume change in gray
and white matter for each participant were then forwarded to
Note: ACC/SMA ¼ anterior cingulate cortex, supplementary motor cortex;
a group analysis, where we compared the changes in volume
rIFG ¼ right inferior frontal gyrus; lSTL ¼ left superior temporal gyrus; AWM ¼
for aerobic exercising and nonaerobic control older adults in
anterior white matter.
FITNESS AND BRAIN HEALTH
49.9. As shown in greater detail in Table 1, groups did not
differ at program onset with respect to average VO2 score,
In this study, we randomly assigned older adult partic-
age, sex, years of education, hormone replacement therapy
ipants to either an aerobic exercise group or a nonaerobic
usage, hypertension, or Mini-Mental State Examination
exercise control group for 6 months and then examined
score. However, after the intervention the aerobically exer-
whether participation in an aerobic exercise regimen would
cising older adults showed a significant increase in VO2.
alter brain volume in an aged cohort. In short, we found that
As predicted, no significant changes in either gray or
participation in an aerobic exercise program increased
white matter volume were detected for our younger par-
volume in both gray and white matter primarily located in
ticipants. However, when directly comparing the changes in
prefrontal and temporal cortices—those same regions that are
gray matter volume for older exercise and control par-
often reported to show substantial age-related deterioration.
ticipants, we found that the previously sedentary aerobic
The current findings are the first, to our knowledge, to
exercising group showed a benefit in brain volume in several
confirm benefits of exercise training on brain volume in aging
regions after participation in an exercise training protocol.
humans. These findings both compliment and extend extant
The blue regions in Figure 1 show areas of gray matter in
human and nonhuman research on the benefits of exercise on
which older adults who participated in the 6-month aerobic
cognition and brain structure such as neuron proliferation and
exercise program showed a significant increase in regional
survival, growth of capillary beds, and increased dendritic
brain volume, compared to older adult controls. As might be
spines (5–13,25). These findings also highlight the potential
expected from the human behavioral research on aerobic
importance of aerobic exercise in not only staving off neural
training effects on cognition (3,4), the largest changes in
decline in aging humans, but also suggest promise as an
volume were present in the frontal lobes of the brain, and
effective mechanism to roll back some of the normal age-
included regions of cortex that are implicated in a broad array
related losses in brain structure (1,23).
of higher order attentional control and memory processes
These results also directly bear on issues of public policy
(20–22). The largest region subsumed portions of the dorsal
and clinical recommendations in that they suggest a rather
anterior cingulate cortex, supplementary motor area, and
simple and inexpensive mechanism to ward off the effects of
middle frontal gyrus bilaterally within the medial walls of the
senescence on human brain tissue. Most importantly, the
brain (ACC/SMA). The second region subsumed a moder-
regions of cortex and white matter that show the greatest
ately large portion of the dorsolateral region of the right
sparing with aerobic fitness play central roles in successful
inferior frontal gyrus, but also part of the posterior aspect of
everyday functioning, and declines in these regions are
the middle frontal gyrus (rIFG), and a third region included
associated with a broad array of clinical syndromes. For
the dorsal aspect of the left superior temporal lobe (lSTL).
example, the prefrontal cortex has been associated with
The yellow region in Figure 1 shows the area in which
critical cognitive processes ranging from inhibitory func-
aerobically exercising participants showed a significant
tioning (22) to measures of general intelligence (26). Losses
increase in white matter volume after the 6-month in-
in this area have been associated with devastating clinical
tervention, compared to control participants. This region was
syndromes such as schizophrenia. The temporal lobes are
in the anterior white matter tracts (AWM), subtending
associated with effective long-term memory function, and
roughly the anterior third of the corpus callosum. Thesewhite matter tracts allow the left and right hemispheres of the
losses in these areas of cortex have been associated with
brain to communicate, and deterioration in these regions has
Alzheimer's dementia in aging populations. Importantly,
been implicated in age-related cognitive decline (23,24). See
these are the same locations that we report brain volume
Table 2 for peak locations, z scores, and cluster sizes.
increases with exercise.
Considering the detrimental impact of age-related brain
These findings, as provocative as they are promising,
volume loss on a broad spectrum of outcomes, it would be
must be viewed with some caution. For example, the older
interesting to investigate the potential for fitness to reduce
adults in our sample were all very healthy and cognitively
the risk of brain tissue loss during the intervention. To
intact. It is not clear whether similar benefits will accrue in
address this issue, we computed a binary outcome measure
pathologically aging individuals. Furthermore, a detailed
of volume change, in which volume loss was coded as
neuropsychological battery was not collected on these
a negative outcome. From this we computed, within each
participants at each time point; therefore, we do not have
cluster reported in Table 2, the relative reduction in risk for
the data to assess how these volumetric changes relate to
brain volume loss associated with participation in the
changes in cognitive scores [but see Erickson and colleagues
aerobic fitness training protocol. Older adults who partic-
(27) for a cross-sectional examination of the relationship of
ipated in the aerobic fitness training protocol showed
fitness-related brain volume differences and cognition]. Our
average reductions in risk, relative to participants in the
relatively small sample size is also a limiting factor. Our
stretching and toning control group, for brain volume loss of
exclusionary criteria limit the interpretation of our results
42.1%, 33.7%, 27.2%, and 27.3%, in the anterior cingulate
to a select group of individuals. Additionally, data from
cortex (ACC/SMA), right superior temporal gyrus (rtSTG),
nonhuman models suggest that the changes in brain volume
right middle frontal gyrus (rtMFG), and anterior white
seen in our study are likely due to changes in synaptic
matter (AWM) clusters, respectively. We should note that
interconnections, axonal integrity, and capillary bed growth,
our sample is somewhat smaller than the recommended
but very little is known about the relationship between the
minimum for risk-reduction estimates, and as such, the risk
voxel-based morphometry methodology used in this study,
reduction estimates should be viewed with some caution.
and the underlying cellular changes that might occur.
COLCOMBE ET AL.
against brain insults of different etiology and anatomy. J Neurosci.
We report the novel and intriguing finding that only 6
11. Niblock MM, Brunso-Berchtold JK, Riddle DR. Insulin-like growth
months of regular aerobic exercise not only spares brain
factor I stimulates dendritic growth in primary somatosensory cortex.
volume but also increases brain volume in an aged cohort.
J Neurosci. 2000;20:4165–4176.
These effects cannot be driven by methodological limi-
12. Churchill JD, Galvez R, Colcombe S, Swain RA, Kramer AF,
tations because neither of the control groups (the older
Greenough WT. Exercise, experience and the aging brain. Neurobiol
nonaerobic exercise participants or the younger control
13. Anderson BJ, Rapp DN, Baek DH, McCloskey DP, Coburn-Litvak PS,
group) showed significant changes in brain volume over 6
Robinson JK. Exercise influences spatial learning in the radial arm
months. Our results suggest that brain volume loss is not an
maze. Physiol Behav. 2000;70:425–429.
inevitable effect of advancing age and that relatively minor
14. Colcombe SJ, Erickson KI, Raz N, et al. Aerobic fitness reduces brain
interventions can go a long way in offsetting and minimi-
tissue loss in aging humans. J Gerontol A Biol Sci Med Sci. 2003;58A:176–180.
zing brain volume loss. Future studies should replicate
15. Karvonen M, Kentala K, Mustala O. The effects of training on heart
these effects using a larger sample size and a more extensive
rate: a longitudinal study. Annales Medicinae Experimentalis et
neuropsychological battery to examine the relationship
Biologiae Fenniae. 1957;35:307–315.
between brain volume changes and cognitive changes.
16. Good CD, Johnsrude IS, Ashburner J, Henson RN, Friston KJ,
Frackowiak RSJ. A voxel-based morphometric study of ageing in 465normal adult human brains. Neuroimage. 2001;14:21–36.
17. Smith SM, Zhang Y, Jenkinson M, et al. Accurate, robust, and
automated longitudinal and cross-sectional brain change analysis.
We thank the National Institute on Aging (RO1 AG25667 and RO1
AG25032) and the Institute for the Study of Aging for supporting this
18. Zhang Y, Brady M, Smith S. Segmentation of brain MR images
through a hidden Markov random field model and the expectation
Address correspondence to Arthur F. Kramer, PhD, Beckman Institute,
maximization algorithm. IEEE Trans Med Imag. 2001;20:45–57.
University of Illinois, 405 N. Mathews Ave., Urbana, IL 61801. E-mail:
19. Friston KJ, Worsley KJ, Frakowiak RSJ, Mazziotta JC, Evans AC.
Assessing the significance of focal activations using their spatial extent.
Hum Brain Map. 1994;1:214–220.
20. Duncan J, Owen AM. Common regions of the human frontal lobe
1. Raz N. Aging of the brain and its impact on cognitive performance:
recruited by diverse cognitive demands. Trends Neurosci. 2000;
integration of structural and functional findings. In Craik F, Salthouse
T, eds. Handbook of Aging and Cognition. Hillsdale, NJ; Erlbaum:
21. Gunning-Dixon FM, Raz N. Neuroanatomical correlates of selected
executive functions in middle-aged and older adults: a prospective
2. Park DC, Polk T, Mikels JA, Taylor SF, Marshuetz C. Cerebral aging:
MRI study. Neuropsychologia. 2003;41:1929–1941.
integration of brain and behavioral models of cognitive function.
22. West R. An application of prefrontal cortex function theory to cognitive
Dialogues Clin Neurosci. 2001;3:151–164.
aging. Psychol Bull. 1995;120:272–292.
3. Colcombe S, Kramer AF. Fitness effects on the cognitive function of
23. O'Sullivan M, Jones DK, Summers PE, Morris RG, Williams SCR,
older adults: a meta-analytic study. Psychol Sci. 2003;14:125–130.
Markus HS. Evidence for cortical ‘‘disconnection'' as a mechanism of
4. Kramer AF, Hahn S, Cohen N, et al. Aging, fitness, and neurocognitive
age-related cognitive decline. Neurology. 2001;57:632–638.
function. Nature. 1999;400:418–419.
24. Colcombe SJ, Kramer AF, Erickson KI, Scalf P. The implications of
5. Black JE, Isaacs KR, Anderson BJ, Alcantara AA, Greenough WT.
cortical recruitment and brain morphology for individual differences
Learning causes synaptogenesis, whereas motor activity causes angio-
in cognitive performance in aging humans. Psychol Aging. 2005;20:
genesis in cerebellar cortex of adult rats. Proc Natl Acad Sci U S A.
25. Trejo JL, Carro E, Torres-Aleman I. Circulating insulin-like growth
6. Rhyu IJ, Boklewski J, Ferguson B, et al. Exercise training associated
factor mediates exercise-induced increases in the number of new
with increased cortical vascularization in adult female cynomologus
neurons in the adult hippocampus. J Neurosci. 2001;21:1628–1634.
monkeys. Abstr Soc Neurosci. 2003;920.
26. Duncan J, Emslie H, Williams P, Johnson R, Freer C. Intelligence and
7. Cotman CW, Berchtold NC. Exercise: a behavioral intervention to
the frontal lobe: the organization of goal-directed behavior. Cognit
enhance brain health and plasticity. Trends Neurosci. 2002;25:
27. Erickson KI, Colcombe SJ, Elavsky S, et al. Interactive effects of
8. van Praag H, Christie BR, Sejnowski TJ, Gage FH. Running enhances
fitness and hormone treatment on brain health in elderly women.
neurogenesis, learning, and long-term potentiation in mice. Proc Natl
Neurobiol Aging. In press.
Acad Sci U S A. 1999;96:13427–13431.
9. Neeper S, Gomez-Pinilla F, Choi J, Cottman C. Exercise and brain
neurotrophins. Nature. 1995;373:109.
Received July 8, 2006
10. Carro E, Trejo LJ, Busiguina S, Torres-Aleman I. Circulating insulin-
Accepted September 21, 2006
like growth factor 1 mediates the protective effects of physical exercise
Decision Editor: Luigi Ferrucci, MD, PhD
Source: http://biologiemartinbolduc.mbolduc1.profweb.ca/wp-content/uploads/2011/09/Colcombe_etal_2006.pdf
ednesday, April 10, 2013 American Conference 17.72. 10. Eric Agee-Floyd ATU 200 100 x 5:07.37. 2. Manuel Alvarez 2. Marcus Lindsey (NLR), 2. Aulexis Pippen (CRO), 26.74. 5. Makayla Daniel Spring Championships, Fullerton, both White Hall, Katie Slaughter and Brit- (LRC), 5:18.18. 3. Christo- 132-5. 3. Charlie Donerson 15.62. 3. Shadeanna Gatlin (NLR), 27.33. 6. Maryma
El campo de copresencia en la estructura conciencia-mundo. Estudio introductorio El campo de copresencia en la estructura con-ciencia-mundo. Estudio introductorio. Jano Arrechea Centro de estudios Parque La Reja, Junio 2010 El campo de copresencia en la estructura conciencia-mundo. Estudio introductorio A. Índice. 1-

