Levitra enthält Vardenafil, das eine kürzere Wirkdauer als Tadalafil hat, dafür aber schnell einsetzt. Männer, die diskret bestellen möchten, suchen häufig nach levitra kaufen ohne rezept. Dabei spielt die rechtliche Lage in der Schweiz eine wichtige Rolle.
Cathelpers.net
J Vet Intern Med 2007;21:1340–1346
Adjuvant Immunother apy of Feline Fibrosarcoma with Recombinant
Feline Interferon-v
Verena Hampel, Bianca Schwarz, Christine Kempf, Roberto Ko¨stlin, Ulrike Schillinger,
Helmut Ku¨chenhoff, Nora Fenske, Thomas Brill, and Johannes Hirschberger
Background: Recombinant feline interferon-v (rFeIFN-v) was tested as a treatment option for cats with fibrosarcoma to
assess safety and feasibility.
Hypothesis: Treatment with rFeIFN-v in cats with fibrosarcoma is safe and feasible.
Animals: Twenty domestic cats.
Methods: In an open-labeled uncontrolled clinical trial 12 injections of 1 3 106 U/kg rFeIFN-v were administered over a 5-
week period: the 1st through 4th injections were given intratumorally, and the 5th through 12th injections were administeredsubcutaneously at the tumor excision site. Wide surgical excision of the tumors was carried out after the 4th injection andbefore the 5th injection of rFeIFN-v. A Common Terminology Criteria for Adverse Events (CTCAE) analysis wasconducted. Flow cytometry of fibrosarcoma cells after incubation with rFeIFN-v and recombinant feline interferon-c wasperformed to assess the biological effect of rFeIFN-v.
Results: Changes in blood cell count, increases in serum aspartate-amino-transferase activity, serum bilirubin concentration,
serum creatinine and serum electrolyte concentrations, weight loss, anorexia, increased body temperature, and reducedgeneral condition were observed but were mostly minor (grade 1 and 2) and self limiting. Eosinophilia (P 5 .025), neutropenia(P 5 .021), and weight loss (P , .001) were statistically correlated with rFeIFN-v-treatment (analysis of parameters beforetreatment and after 3 injections of rFeIFN-v). Flow cytometry of 5 unrelated feline fibrosarcoma cell lines showed increasedexpression of major histocompatibility complex (MHC) class I molecules (P 5 .026) in response to in vitro incubation withrFeIFN-v, whereas expression of MHC class II molecules was not affected significantly.
Conclusions and Clinical Importance: RFeIFN-v for the treatment of feline fibrosarcoma is safe, well tolerated, and can be
easily performed in practice. To assess the efficacy of the treatment, it should be tested in a placebo-controlled trial.
Key words: Antitumor immunity; Cat; Cytokines; Major histocompatibility complex; Soft-tissue sarcoma.
Fibrosarcomas are common in the cat and comprise free interval or reduce tumor recurrence or the rate of
over 40% of all skin tumors in this species.1 Because
of the invasive nature of fibrosarcomas, the recurrence
Cancer immunotherapy refers to using the power and
rate ranges from 30 to 70%,2,3 and metastasis occurs in
the specificity of the immune system for the treatment of
approximately 10 to 20% of affected cats.4–7 Currently,
a malignancy. This therapy is based on the use of
there is no successful treatment for fibrosarcoma in the
biologically active proteins with the aim of altering the
cat.7 Although surgery is the treatment of choice,
specific and nonspecific immune responses of the
complete excision often is difficult. Wide surgical
patient. The immune system is capable, under certain
excision extends the tumor-free interval and increases
circumstances, of recognizing and eliminating tumor
survival time.8,9
Pre- or postoperative radiation therapy extends the
Interferons (IFN) are cytokines that have antiviral,
median tumor-free time up to 422 days10 and may
antiproliferative, and immunomodulatory effects. The
reduce the recurrence rate to 42% in cats with clean
family of IFNs consists of 2 major classes, type I and type
surgical margins.11 In the study by Cohen et al,4
II. Type I IFNs include IFN-a, b, d, t, and v, whereas
a recurrence rate of 41% after tumor excision, elec-
IFN-c is the only type II IFN. The biological activities of
tron-beam radiation, and, in some cases, chemotherapy
type I IFNs include growth inhibition of tumor cells,16,17
was documented. Chemotherapy regimens that have
induction of apoptosis,17,18 natural killer cell activation,19
been investigated include doxorubicin12,13 and carbopla-
and an increase in the expression of major histocompat-
tin.11 Martano et al14 showed that the combination of
ibility complex (MHC) class I molecules.20,21 The amino
surgery and doxorubicin does not increase the disease-
acid sequence of recombinant feline IFN-v has approx-imately 60% homology to that of human IFN-v.22 The
From the Department of Veterinary Internal Medicine, Veteri-
antitumor efficacy of human IFN-v has been described in
nary Faculty, LMU Munich, Munich, Germany (Hampel, Schwarz,
vitro23,24 and in vivo.25 Recombinant feline IFN-v has
Kempf, Hirschberger); Department of Veterinary Surgery, Veteri-
approximately 60% homology to human IFN-a.22,26
nary Faculty, LMU Munich, Munich, Germany (Ko¨stlin); Institute
Human IFN-a has been used in Germany since 1999
for Experimental Oncology, Klinikum Rechts der Isar, TU Munich,
for the adjuvant therapy of malignant melanoma in
Munich, Germany (Schwarz, Kempf, Schillinger, Brill); and the
humans. Because of the close relationship of IFN-v and
Statistical consulting unit, Department of Statistics, LMU Munich,
IFN-a, adjuvant immunotherapy of feline fibrosarcoma
Mu¨nchen, Germany (Ku¨chenhoff, Fenske).
by using rFeIFN-v follows the therapy protocol for
Reprint requests: Verena Hampel, Mangstrasse 7, D-80997
human IFN-a.27 Recombinant feline IFN-a is not
Munich, Germany; e-mail:
[email protected].
commercially available, whereas rFeIFN-v is the only
Submitted August 6, 2006; Revised December 12, 2006, March 3,
IFN licensed for use in cats.
2007, April 3, 2007; Accepted June 19, 2007.
IFN-c increases antigen presentation by upregulation
Copyright E 2007 by the American College of Veterinary Internal
of MHC expression on antigen presenting cells and by
activation of natural killer cells.21,28,29 Tumor cells often
Fibrosarcoma Therapy with Interferon-v
have a decreased expression of MHC class I mole-
through 4th injection) and then subcutaneously (SC) (5th through
cules.15,30–33 This so-called tumor escape mechanism
12th injection) at the tumor excision site. In the first week of
prevents recognition of tumor antigens by cytotoxic T
treatment, the cats received 1 intratumoral injection daily for 3
cells, because tumor antigens are presented by MHC
consecutive days. In the second week, the cats again received 1
class I molecules on the surface of malignant cells.
injection daily for 3 consecutive days, but the 4th injection wasadministered intratumorally and the 5th and 6th injections were given
Because IFN-v and IFN-c may be able to increase
SC at the excision site after surgical excision of the tumor. In the next
expression of MHC class I molecules and, therefore,
3 weeks, the cats received 2 injections of rFeIFN-v SC per week.
antigen presentation, these cytokines may render tumorcells susceptible to attack by natural killer cells,cytotoxic T cells, activated macrophages, or antibo-
dies.32,34 To assess the biological effect of rFeIFN-v on
Wide surgical excision (ie, tumor excision en bloc with 3-cm
MHC class I and II expression on feline fibrosarcoma
margins to palpable evidence of disease) was carried out during the
cells, flow cytometry from 5 unrelated fibrosarcoma cell
second week of treatment, after the 4th and before the 5th injection
lines was carried out. The effect of rFeIFN-v was
of rFeIFN-v. All of the operations were performed by the same
compared with that of rFeIFN-c. RFeIFN-v was
surgeon (RK). Cross-sectional imaging was not performed in any
administered to domestic cats before and after tumor
excision. To evaluate the safety and the feasibility of thisadjuvant treatment, an open-label prospective clinical
Pain Relief and Antibiotic Treatment
trial was conducted in 20 cats.
For perioperative analgesia, buprenorphineb was given at
a dosage of 0.01 mg/kg on the day of surgery. For postoperative
Materials and Methods
analgesia, all cats received meloxicamc for 5 days (1st day aftersurgery: 0.3 mg/kg; 2nd through 5th days after surgery: 0.1 mg/kg).
Furthermore, the cats were treated with amoxicillin-clavulanicacidd at a dosage of 25 mg/kg per day for 5 days.
The study was an open-label, single-arm, monocentric, pro-
spective, safety and feasibility study. From January 2004 untilAugust 2004, cats with fibrosarcoma were evaluated for inclusion
in the study. Cats were enrolled in the study if they met definedinclusion criteria: resectable tumor (excision possible without
The cats were reevaluated for tumor recurrence on days 28, 42,
opening a visceral cavity, amputation or partial resection of parts
90, 180, 270, and 360 after surgery or on an ad hoc basis if the
of the scapula or the spine), absence of previous immunomodu-
owner reported regrowth of the tumor. At each reevaluation, the
latory treatment within 6 weeks of the start of IFN-v treatment,
cats underwent a clinical examination, which included weighing, as
and life expectancy .1 year. Twenty cats with fibrosarcoma met
well as evaluation of wound healing, potential adverse effects,
the inclusion criteria: 18 Domestic Shorthair, 1 Domestic Longhair
potential recurrence, and metastasis of the tumor. On days 28, 42,
and 1 Persian cat. They ranged in age from 4 to 16 years (mean,
180, and 360, CBC count and serum biochemistry were performed.
10 years), and there were 11 castrated male and 9 spayed female
The final evaluation on day 360 also included thoracic (left and
cats. Eleven cats had primary tumors, and 9 cats had tumors that
right lateral and ventrodorsal views) and abdominal (lateral view)
had recurred after surgery.
radiographs and abdominal ultrasonography.
The tumors were mainly located in the interscapular region (11
cats). Other sites were the thoracic wall (5 cats), the lumbar area (3cats), and the shoulder region (1 cat). The tumors all were located
Common Terminology Criteria for Adverse Events
at sites that are commonly used for administration of vaccines.
All data were recorded in detail in case report forms and were
However, it was not possible to determine whether vaccines had
analyzed to determine the toxicity of the treatment. To provide
indeed been given at the tumor site. Nine cats had stage I tumors
a basis for the evaluation of toxicity, the ‘‘Veterinary Co-operative
(,2 cm), 8 cats had stage II tumors (2–3 cm), and 3 cats had stage
Oncology Group—Common terminology criteria for adverse
III tumors (.3 cm). Two cats had multiple tumors at 1 site. In both
events following chemotherapy or biological antineoplastics
cats, 3 tumors were present at 1 site. In these cats, the diameters of
therapy in dogs and cats'' V 1.0 was used.37
the tumors were added (tumor staging according to Hirschbergerand Kessler35). Nine cats could not be included in the study becauseof metastasis in the liver (1 cat), nonresectable tumor (4 cats), no
clinical evidence of fibrosarcoma (3 cats), and 2 tumors at 2different sites (1 cat).
Five unrelated feline fibrosarcoma cell lines (fibrosarcomas
from control animals of another study) were cultured at theInstitute for Experimental Oncology, TU Munich, Munich,
Reagents and Products
Germany, by standard methods: cells were mechanically andenzymatically dissociated and cultured in Dulbecco's Modified
Recombinant FeIFN-va was provided by Virbac, S.A., France.
Eagle Mediume supplemented with 10% fetal calf serum,f 1%
The 5 3 106 U powder fraction was diluted in 1 ml of isotonic
glutamine,g 100 U/mL penicillin,h and 100 mg/mL streptomycini at
saline solution immediately before use.
37uC in a 5% CO2 atmosphere.
Immunotherapy with rFeIFN-v
Incubation with rFeIFN-v and rFeIFN-c
The treatment protocol consisted of 12 injections of 1 3 106 U/
kg rFeIFN-va over 5 weeks. The dosage of 1 3 106/kg rFeIFN-v
Fibrosarcoma cells lines (1.75 3 106 cells each) were incubated
has been established in cats and is recommended by the
in vitro for 48 hours with 50 mL isotonic saline solution (negative
manufacturer.36 RFeIFN-v was administered intratumorally (1st
control), 1,000 ng rFeIFN-c,j or 25,000 U rFeIFN-v.a
Staining of the Cells
Modulation of MHC Molecules on Fibrosarcoma Cells
by rFeIFN-v and rFeIFN-c
The flow cytometry protocol described by Schwarz28 was used.
For each tumor cell line, 13 FACS tubesk were prepared (3–4 3 105
Expression of MHC class I and II antigens on feline
cells each): 3 for negative control, 5 for each rFeIFN-v and
fibrosarcoma cell lines was evaluated by using flow
rFeIFN-c. The monoclonal antibodies used were mouse anti-MHC
cytometry (see Fig 2). A 1-fold increase of expression of
class I v6/32l and mouse anti-feMHC class II 42.3.m The antibodies
MHC class I molecules was observed in fibrosarcoma
were added as follows: (a) no antibody (negative control), (b) 10 mL
cell lines in response to in vitro incubation with
immunogloublin (Ig) G1,n and (c) 10 mL IgG2ao (isotype controls),(d) 60 mL MHC I antibody (v6/32), and (e) 10 mL MHC II
25,000 U rFeIFN-v (P 5 .026). The expression of
antibody (42.3). The cells were washed twice and incubated with
MHC class II molecules also was influenced by rFeIFN-
10 mL fluorescein isothiocyanate-conjugated rabbit anti-mouse
v, but this effect was not statistically significant (P 5
IgG antibodyp for 20 minutes in the dark and resuspended
.22). The incubation of feline tumor cell lines with
1,000 ng rFeIFN-c caused a 1.5-fold increase ofexpression of MCH class I (P 5 .010) and a 3-foldincrease of expression of MHC class II molecules (P 5
Flow Cytometric Analysis
.020). This observation means that rFeIFN-v is able to
Expression of MHC class I and II on the cell surface was
increase the expression of MHC class I molecules on
evaluated by performing flow cytometric analysis with FACS-
feline fibrosarcoma cells, whereas rFeIFN-c can mod-
Vantage.q Negative and isotype controls were used as previously
ulate the expression of MHC class I and II molecules.
described.28 The Cell Quest Programr was used for data analysis.
Statistical Analysis
Immunotherapy has been investigated in several
Kaplan-Meier analysis of recurrence was carried out by using
studies in cats with fibrosarcoma. King et al38 reported
SPSS v.13.Os. The number of events (3 in the group of cats without
that the immunostimulant acemannan may be an
previous recurrence, 6 in the group of cats with at least 1 previous
effective adjunct to surgery and radiation therapy in
recurrence) was too small for additional statistical analyses. A
the treatment of fibrosarcoma. Acemannan enhances
paired Student's t-test was used to evaluate differences between the
macrophage release of interleukin-1, interleukin-6,
serum results and blood counts of days –7 (before 1st treatment)
tumor necrosis factor-a, and IFN-c. Jourdier et al39
and 0 (day before surgery) and to test for change of expression ofMCH class I and II. In all statistical analyses, a P value of ,.05
studied local immunotherapy of spontaneous feline
was considered significant.
fibrosarcomas by using recombinant poxviruses expres-sing interleukin-2. This study demonstrated a decrease intumor recurrence rates from 61% (11 of 18 cats) in
control animals (treated with surgery and iridium-basedradiotherapy) to 39% (7 of 18) and 28% (5 of 18) in cats
Common Terminology Criteria for Adverse Events
who received human or feline interleukin-2 in addition
The Common Terminology Criteria for Adverse
to surgery and iridium-based radiotherapy.
Events (CTCAE) analysis produced the results shown
Recombinant feline IFN-v has antiviral40,41 and
in Table 1. The statistical analysis of CBC count, serum
antiproliferative effects,42,43 and has been used in the
biochemistry results, and body weight on days –7
treatment of feline calicivirus infection44; feline herpetic
(before treatment) and 0 (after 1 week of treatment
keratitis45; feline leukemia virus (FeLV) and feline
but before surgery) produced the following results: the
immunodeficiency virus (FIV) infection46; feline infec-
parameters neutropenia (P 5 .021), eosinophilia (P 5
tious peritonitis47; gingivitis, stomatitis, and orophar-
.025), and weight loss (P , .001) showed significance
yngitis48; and canine parvovirus infection.49 De Mari et
and were positively correlated with the rFeIFN-v
al46 tested rFeIFN-v in 81 cats with FeLV and with
FeLV and FIV-coinfection. In this placebo-controlledtrial, the cats were treated with 1 3 106 U/kg rFeIFN-vfor 5 consecutive days in 3 series on day 0, day 14, and
Clinical Findings
day 60. The investigators observed systemic adverse
The tumor recurred locally in 9 of 20 cats (45%); 1
events in 5 of 81 of the cats, including lethargy, transient
other cat developed pulmonary metastases and died on
vomiting, and diarrhea, but with no statistically
day 258. The tumor had not recurred at the original site.
significant differences between the IFN-treated group
The disease-free interval of this cat was censored at day
and the placebo group.
258, but it was included in the statistical analysis. At the
In another clinical trial,48 20 cats with chronic
final evaluation, on day 360, 10 cats were disease free.
gingivitis, stomatitis, or oropharyngitis were treated by
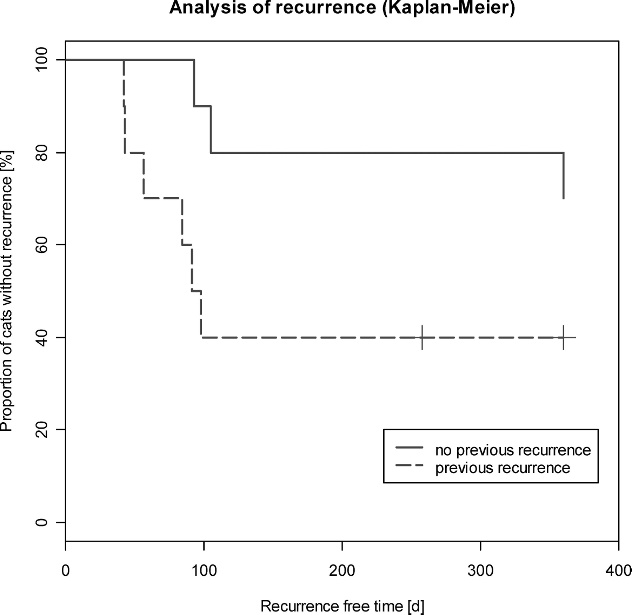
To investigate the influence of previous recurrences on
using subgingival and SC injections of 1 3 106 U/kg or
recurrence-free time, the study population was divided
2.5 3 106 U/kg rFeIFN-v. Mihaljevic48 found transient
into 2 groups: a group without previous recurrence (10
lethargy, increased body temperature, and anorexia in
cats) and a second group with at least 1 previous
some cats. These adverse events were mainly observed in
recurrence (10 cats). The Kaplan-Meier curves for both
cats treated at a dosage of 2.5 3 106 U/kg. In the present
groups are shown in Fig 1.
study, cats were treated at a dosage of 1 3 106 U/kg, as
Fibrosarcoma Therapy with Interferon-v
Number of adverse events during the study according to the Veterinary Co-operative Oncology Group
Adjuvant Treatment
(28 adverse events)
(53 adverse events)
(101 adverse events)
(68 adverse events)
28 (last treatment
Segmented neutropenia 3
Wound healing disorder
q, increased parameter; Q, decreased parameter.
a From Ref. 37.
recommended by the manufacturer36 to avoid the above-
weight loss, which indicated a correlation to the
mentioned adverse events of the higher dosage.
treatment with rFeIFN-v. The manufacturer36 describes
The results of the CTCAE analysis of the study
neutropenia during treatment with rFeIFN-v. Mild
correspond with the adverse effects specified by the
eosinophilia may be induced by rFeIFN-v. Weight loss
manufacturer.36 These included hyperthermia; vomiting;
showed a clear statistical significance (P , .001), which
diarrhea; mild decrease in the total number of leuko-
could be correlated to the therapy or stress caused by
cytes, thrombocytes and erythrocytes; increased activity
frequent visits in the clinic. Weight loss was not observed
of serum aspartate-amino-transferase; and transient
in the above-mentioned studies.46,48
lethargy. However, these changes generally were minor
In the present study, there was a recurrence rate of
grade and self limiting. The statistical analysis of blood
45% (9 of 20 cats); metastases were found in 1 cat. The
test results and body weight on days –7 (before
current study design was not able to a demonstrate
treatment) and 0 (after 1 week of treatment) showed
decreased recurrence rate, because of a lack of a control
significant differences in neutropenia, eosinophilia, and
group and a lack of surgical margin assessment after
study, cats were enrolled in the trial as they werepresented in the clinic, therefore, 17 cats randomly hadtumor sizes of ,3 cm. This observation can beattributed to the awareness of the referring veterinariansabout fibrosarcomas.
The cats had to meet defined inclusion criteria: 4 cats
were not included in the study, because they hadnonresectable tumors. In these cats, tumor masses wereso large that tumor excision was not possible withoutopening a visceral cavity or performing amputation orpartial resection of parts of the scapula or the spine.
Cats with 2 tumors at 2 different sites (eg, thoracic
wall and lumbar area) could not be included in thestudy. It cannot be assumed that 2 tumors at 2 differentsites originated from the same tumor cells. Two differentfibrosarcomas might not have the same biologicalbehavior and potentially may have different antigenicproperties. The cats received recombinant feline IFN-vintratumorally or SC at the excision site, and for thisreason only 1 tumor could be treated. Consequently, 1cat with tumors at different sites was excluded from the
Analysis of recurrence (Kaplan-Meier) of cats without
study. Two cats had multiple tumors at 1 site. In both
previous recurrences (n 5 10) and cats with at least 1 previous
cats, 3 tumors were present at 1 site, for 1 cat, it was the
recurrence (n 5 10).
first recurrence and, for the other cat, it was the secondrecurrence. This finding was thought to represent
surgery. Histologic grading of the surgical specimens as
a recurrence of 1 fibrosarcoma as multiple small nodules
described previously was not performed.
at the surgical site.
Tumors in the study described here all were located at
Tumors were resected en bloc with 3-cm margins to
vaccination sites. However, it was not possible to
palpable evidence of disease, and tumor excisions always
determine whether vaccines had indeed been given at
were carried out by the same experienced surgeon. Cohen
the tumor site. Hendrick et al50 reported that 39% of cats
et al4 stated that the location and the size of the primary
with vaccination site sarcomas had tumor sizes of 2.0–
tumor and the ability to remove it are important
3.9 cm and 33% of the cats had tumor sizes of 4.0–
prognostic factors. The recurrence rate in the present
5.0 cm. However, 33% of cats with nonvaccination site
study could have been influenced by the fact that most of
sarcomas had tumor sizes of 2.0–3.9 cm. In the present
the cats had small tumors and that the surgical procedurewas performed by an experienced surgeon.
Cohen et al4 also reported that the prognosis of
recurrent fibrosarcoma treated by surgical excision isworse than that of excision of primary fibrosarcoma. Inthe present study, the number of tumor recurrencesbefore rFeIFN-v treatment also may have had animpact on the outcome. Seven cats that had tumorrelapses (6) or metastases (1) had a minimum of 1fibrosarcoma removed surgically before inclusion in thestudy. Of these 7 cats, 4 had 1 previous fibrosarcomaand 3 had 2 previous tumors. In contrast, only 3 catswith a primary tumor had tumor recurrence in thisstudy. Of the 10 cats that were tumor free at the end ofthe study, 7 had a primary tumor and 3 had had 1previous fibrosarcoma at the time of admission. Visual
Results of flow cytometry of feline fibrosarcoma cell lines
inspection of the Kaplan-Meier curve (Fig 1) indicated
incubated in vitro with 25,000 U (rFeIFN-v) and 1,000 ng
an influence of previous recurrences on recurrence-free
rFeIFN-c for 48 hours. The figure shows 1 of 5 experiments
time, but, presumably because of small sample sizes, this
representative of all 5 fibrosarcoma cell lines tested. The y-axis
effect did not achieve statistical significance.
shows the counts, and the x-axis shows the intensity of the
All of the cats received meloxicam for postoperative
fluorescence of fluorescein isothiocyanate-conjugated rabbit anti-
pain once daily for 5 days, as described in other
mouse immunogloublin (Ig) G antibody (FITC). 2.1.: solid line:
studies.51,52 Nonsteroidal anti-inflammatory drugs also
major histocompatibility complex (MHC) class I after stimulation
have been beneficial in the treatment of transitional-cell
with rFeIFN-c, dotted line: negative control; 2.2.: solid line: MHC
carcinoma in dogs.53 These substances not only decrease
class II after stimulation with rFeIFN-c; 2.3.: solid line: MHC classI after stimulation with rFeIFN-v; 2.4.: solid line: MHC class II
the inflammatory reaction in the tumor tissue but also
after stimulation with rFeIFN-v.
inhibit tumor growth. It seems unlikely that meloxicam
Fibrosarcoma Therapy with Interferon-v
retarded tumor growth in this study, because it was
Faculty and Prof Dr Walter Hermanns, Department of
administered for only 5 days.
Veterinary Pathology of the Veterinary Faculty, LMU
The expression of MHC class I and II antigens on
Munich, Munich, Germany, and Edelburga Ham-
feline fibrosarcoma cell lines was evaluated by using
merschmid and Gerlinde Stejskal from the Institute for
flow cytometry. An increase in expression of MHC class
Experimental Oncology, Klinikum Rechts der Isar, TU
I molecules was observed in all tested fibrosarcoma cell
Munich, Munich, Germany. The trial was funded by
lines after in vitro incubation with recombinant feline
Virbac, Carros, France.
IFN-v. The expression of MHC class II molecules wasaffected by rFeIFN-v to a lesser extent. The incubation
of feline tumor cell lines with rFeIFN-c resulted inincreased cellular expression of MCH class I and II
1. Stiglmair-Herb MT. Hauttumoren bei Katzen—Eine retro-
molecules. IFN-v is a type I IFN and is able to increase
spektive studie. Tiera¨rztl Umschau 1987;54:681–686.
the expression of MHC class I molecules.19,20 In contrast,
2. Macy DW, Couto CG. Review of treatment options for
IFN-c is able to modulate the expression of MHC class I
vaccine-associated feline sarcoma. J Am Vet Med Assoc 1998;
and II antigens. This effect has been described in
different tumor cell lines in vitro.21,28,29 An increase in
3. McEntee MC, Page RL. Feline vaccine-associated sarcomas.
the expression of MHC class I antigens, and, therefore,
J Vet Intern Med 2001;15:176–182.
4. Cohen M, Wright JC, Brawner WR, et al. Use of surgery and
an increase in antigen presentation, render tumor cells
electron beam irradiation, with or without chemotherapy, for
susceptible to attack by the immune system.32,34 Re-
treatment of vaccine-associated sarcomas in cats: 78 cases (1996–
cently, Murgia et al33 reported down-modulation of
2000). J Am Vet Med Assoc 2001;219:1582–1589.
MHC antigen expression in a canine transmissible
5. Esplin DG, McGill LD, Meininger AC, et al. Postvaccina-
venereal tumor as a tumor escape mechanism. There-
tion sarcomas in cats. J Am Vet Med Assoc 1993;202:1245–1247.
fore, by increasing cellular expression of MHC I,
6. Hershey AE, Sorenmo KU, Hendrick MJ, et al. Prognosis
recombinant feline IFN-v may promote tumor-cell
for presumed feline vaccine-associated sarcoma after excision: 61
destruction by the immune system. Immunotherapy
cases (1986–1996). J Am Vet Med Assoc 2000;216:58–61.
with recombinant feline IFN-v in cats with fibrosarco-
7. Macy DW, Couto CG. Prevention and treatment of in-
ma is safe and well tolerated.
jection-site sarcomas. J Feline Med Surg 2001;3:169–170.
The treatment is straightforward and can be per-
8. Davidson EB, Gregory C, Kass PH. Surgical excision of soft
formed easily in clinical practice. The results of the
tissue fibrosarcomas in cats. Vet Surg 1997;26:265–269.
present study encourage placebo-controlled clinical
9. Dillon CJ, Mauldin GN, Baer KE. Outcome following
trials to evaluate the efficacy of this treatment.
surgical removal of nonvisceral soft tissue sarcomas in cats: 42cases (1992–2000). J Am Vet Med Assoc 2005;227:1955–1957.
10. Steger-Lieb A, Kostorz A, Hauser B, et al. Einsatz der
Strahlentherapie beim vakzineassoziierten Sarkom der Katze.
Erfahrungen aus 18 Fa¨llen (1994–1999). Tiera¨rztl Prax 2002;30:35–40.
a Virbagen Omega, Batch no OU4M,
11. Kobayashi T, Hauck ML, Dodge R, et al. Preoperative
b Temgesic, Essex Pharma, Munich, Germany
radiotherapy for vaccine associated sarcoma in 92 cats. Vet Radiol
c Metacam, Boehringer Ingelheim, Ingelheim, Germany
d Synulox, Pfizer, Karlsruhe, Germany
12. Barber LG, Sorenmo KU, Cronin KL, et al. Combined
e DMEM, Biochrom, Berlin, Germany
doxorubicin and cyclophosphamide chemotherapy for nonresect-
f FKS, Biotech, Aidenbach, Germany
able feline fibrosarcoma. J Am Anim Hosp Assoc 2000;36:416–421.
g Glutamine, Biochrom, Berlin, Germany
13. Poirier VJ, Thamm DH, Kurzman ID, et al. Liposome-
h Penicillin, Biochrom, Berlin, Germany
encapsulated doxorubicin (Doxil) and doxorubicin in the treatment
i Streptomycin, Biochrom, Berlin, Germany
of vaccine-associated sarcoma in cats. J Vet Intern Med
j Recombinant Feline Interferon-y, R&D Systems, Wiesbaden,
14. Martano M, Morello E, Ughetto M, et al. Surgery alone
k FACS tubes, BD, Pharmingen, Germany
versus surgery and doxorubicin for the treatment of feline
l wb/32, Abcam, Cambridge, United Kingdom
injection-site sarcomas: A report on 69 cases. Vet J 2005;170:84–90.
m 42.3, Serotec, Oxford, UK
15. Blattman JN, Greenberg PD. Cancer immunotherapy: A
n IgG1, BD, Pharmingen, Germany
treatment for the masses. Science 2004;305:200–205.
o IgG2a, BD, Pharmingen, Germany
16. Otsuki T, Yamada O, Sakaguchi H, et al. Human myeloma
p FITC, DAKO Diagnostica, Hamburg, Germany
cell apoptosis induced by interferon-alpha. Br J Haematol
q FACS-Vantage, Becton-Dickinson, San Jose, CA
r Cell Quest Program, Becton-Dickinson, San Jose, CA
17. Baldwin SL, Powell TD, Sellins KS, et al. The biological
s SPSS, Chicago, Illinois
effects of five feline IFN-a subtypes. Vet Immunol Immunopathol2004;99:153–167.
18. Beatu TM, Hiscott J. On the TRAIL to apoptosis. Cytokine
Growth Factor Rev 2002;13:199–207.
19. Taki S. Type I interferons and autoimmunity: Lessons from
the clinic and from IRF-2-deficient mice. Cytokine Growth FactorRev 2002;13:379–391.
The authors thank the anesthesia team from the
20. Stark GR, Kerr IM, Williams BR, et al. How cells respond
Department of Veterinary Surgery of the Veterinary
to interferons. Annu Rev Biochem 1998;67:227–264.
21. Yang I, Kremen T, Giovannone A, et al. Modulation of
radiation therapy on spontaneous canine and feline fibrosarcomas.
major histocompatibility complex class I molecules and major
J Am Anim Hosp Assoc 1995;31:439–447.
histocompatibility complex-bound immunogenic peptides induced
39. Jourdier TM, Moste C, Bonnet MC, et al. Local immuno-
by interferon-a and interferon-c treatment of human glioblastoma
therapy of spontanoues feline fibrosarcomas using recombinant
multiforme. J Neurosurg 2004;100:310–319.
poxviruses expressing interleukin 2 (IL2). Gene Ther 2003;10:
22. Ueda Y, Sakurai T, Yanai A. Homogeneous production of
feline interferon in silkworm by replacing single amino acid code in
40. Truyen U, Blewaska S, Schultheiss U. Untersuchung der
signal peptide region in recombinant baculovirus and character-
antiviralen Wirksamkeit von Interferon-Omega gegen ausgewa¨hlte
ization of the product. J Vet Med Sci 1993;55:251–258.
Viren von Hund und Katze. Prakt Tierarzt 2002;83:862–865.
23. Nieroda C, Pestka S, Schlom J, et al. Interferon-v augments
41. Siebeck N, Hurley DJ, Garcia M, et al. Effects of human
major histocompatibility and human tumor-associated antigen
recombinant alpha-2b interferon and feline recombinant omega
expression. Mol Cell Differ 1996;4:335–351.
interferon on in vitro replication of feline herpesvirus-1. Am J Vet
24. Tiefenthaler M, Geisen F, Schirmer M, et al. A comparison
of the antiproliferative properties of recombinant human IFN-a2
42. Priosoeryanto BP, Tateyama S, Yamaguchi R, et al.
and IFN-v in human bone marrow culture. J Interferon Cytokine
Antiproliferation and colony-forming inhibition activities of
recombinant feline interferon on various cells in vitro. Can J Vet
25. Horton HM, Hernandez P, Parker SE, et al. Antitumor
effects of interferon-v: In vivo therapy of human tumor xenografts
43. Tateyama S, Priosoeryanto BP, Yamaguchi R, et al. In vitro
in nude mice. Cancer Res 1999;59:4064–4068.
growth inhibition activities of recombinant feline interferon on all
26. Nakamura N, Sudo T, Matsuda S, et al. Molecular cloning
lines derived from canine tumors. Res Vet Sci 1995;59:275–277.
of feline interferon cDNA by direct expression. Biosci Biotech
44. Gaskell R, Dawson S. Feline respiratory disease. In: Greene
C, ed. Infectious Diseases of the Dog and Cat. 2nd ed. Philadephia,
27. Pirard D, Heenen M, Melot C, et al. Interferon alpha as
PA: WB Sanders; 1998:97–106.
adjuvant postsurgical treatment of melanoma: A meta analysis.
45. Verneuil M. Topical application of feline interferon omega
in the treatment of herpetic keratitis in the cat: Preliminary study.
28. Schwarz B. Klonieren der Felinen Zytokin-Gene IL-2, GM-
Proceedings of European College of Veterinary Opthalmologists,
CSF und IFN-c zum Adjuvanten, Nonviralen Gentherapeutischen
Munich, Germany, June 2004.
Einsatz beim Fibrosarkom der Katze. Mu¨nchen, Germany: Uni-
46. De Mari K, Maynard L, Sanquer A, et al. Therapeutic
versita¨t Mu¨nchen; 2005. Diss med vet.
effects of recombinant feline interferon-v on feline leukemia virus
29. Whitley E, Bird A, Zucker K, et al. Modulation by canine
(FeLV)-infected and FeLV/feline immunodeficiency virus (FIV)-
interferon-gamma of major histocompatibility complex and tumor-
coinfected symptomatic cats. J Vet Intern Med 2004;18:477–482.
associated antigen expression in canine mammary tumor and
47. Ishida T, Shibancia A, Tonaha S, et al. Recombinant feline
melanoma cell lines. Anticancer Res 1995;15:923–929.
interferon therapy of feline infectious peritonitis. Proceedings of
30. Melero I, Bach N, Chen L. Costimulation, tolerance and
the 2nd International Feline Coronavirus (FcoV)/Feline Infectious
ignorance of cytolytic T-lymphocytes in immune responses totumor antigens. Life Sci 1997;60:2035–2041.
Peritonitis (FIP)-Symposium, Glasgow, UK, August 2002:17,
31. Seliger B, Cabrera T, Garrido F, et al. HLA class I antigen
48. Mihaljevic S. First clinical experiences with omega-in-
abnormalities and immune escape by malignant cells. Semin
terferon in the treatment of chronic gingivitis-stomatitis-orophar-
Cancer Biol 2002;12:3–13.
yngitis of cats. Prakt Tierarzt 2003;84:350–361.
32. Elmslie RE, Dow SW. Genetic immunotherapy for cancer.
49. De Mari K, Maynard L, Eun H, et al. Treatment of canine
Semin Vet Med Surg (Small Anim) 1997;12:193–205.
parvoviral enteritis with interferon-omega in a placebo-controlled
33. Murgia C, Pritchard JK, Kim SY, et al. Clonal origin and
field trial. Vet Rec 2003;152:105–108.
evolution of a transmissible cancer. Cell 2006;126:477–487.
50. Hendrick MJ, Shofer FS, Goldschmidt MH, et al.
34. Blaese M, Blankenstein T, Brenner M, et al. European
Comparison of fibrosarcomas that developed at vaccination sites
School of Oncology position paper. Gene therapy for the medical
and at nonvaccination sites in cats: 239 cases (1991–1992). J Am
oncologist. Eur J Cancer 1995;31A:1531–1537.
Vet Med Assoc 1994;205:1425–1429.
35. Hirschberger J, Kessler M. Das feline Fibrosarkom.
51. Slingsby L, Waterman-Pearson A. Comparison between
Tiera¨rztl Prax 2001;29:66–71.
meloxicam and carprofen for postoperative analgesia after feline
36. Virbac. Product information Virbagen Omega. Virbac 2001.
ovariohysterectomy. J Small Anim Pract 2002;43:286–289.
37. Vail D. Veterinary co-operative oncology group—Common
52. Robertson S, Taylor P. Pain management in cats—Past,
terminology criteria for adverse events (VCOG-CTCAE) following
present and future. Part 2. Treatment of pain—clinical pharma-
chemotherapy or biological antineoplastic therapy in dogs and cats
cology. J Feline Med Surg 2004;6:321–333.
v1.0. Vet Comp Oncol 2004;2,4:194–213.
53. Knapp D, Richardson R, Chan T, et al. Piroxicam therapy
38. King GK, Yates KM, Greenlee PG, et al. The effect of
in 34 dogs with transitional cell carcinoma of the urinary bladder.
acemannan immunostimulant in combination with surgery and
J Vet Intern Med 1994;8:273–278.
Source: http://www.cathelpers.net/wp-content/uploads/2011/12/felinefibrosarcoma.1.pdf
Carbamate and Pyrethroid Resistance in the Leafminer J. A. ROSENHEIM,' AND B. E. T ABASHNIK Department of Entomology, University of Hawaii at Manoa, Honolulu, Hawaii 96822 J. Econ.Entomol.83(6): 2153-2158 (1990) ABSTRACT Populations of D1glyphus begini (Ashmead), a parasitoid of Lirlomyza leafminers, showed resistance to oxamyl, methomyl, fenvalerate, and permethrin in labo-ratory bioassays. Relative to a susceptible strain from California, maximum resistance ratiosfor these pesticides were 20, 21, 17, and 13, respectively. Three populations that had beentreated frequently with insecticides were significantly more resistant to all four insecticidescompared with an untreated Hawaii population and a California population with an unknownspray history. Parasitoids from a heavily sprayed tomato greenhouse on the island of Hawaiihad LC",'s for permethrin and fenvalerate that were 10 and 29 times higher than the fieldrate, respectively. Populations resistant to oxamyl and methomyl had LC",'s two- and sixfoldbelow the field rate, respectively. D. begini is one of the few parasitoids resistant to pyre-throids, with LC",'s exceeding field application rates. Resistant D. begini may be useful forcontrolling leafminers in management programs that integrate biological and chemical con-trols.
AFRICAN AMERICAN DIET by Mankind Diet & Health International, On the web at: http://www.aadiet.net Copyright 2005, 2006 All rights reserved. For information about permission to reproduce selections from this book, send your email request to: [email protected] or The contents of this book are meant as an opinion of how to lose weight and live a healthy active life. It is NOT a replacement or substitute for

