Levitra enthält Vardenafil, das eine kürzere Wirkdauer als Tadalafil hat, dafür aber schnell einsetzt. Männer, die diskret bestellen möchten, suchen häufig nach levitra kaufen ohne rezept. Dabei spielt die rechtliche Lage in der Schweiz eine wichtige Rolle.
Ictopen2016_dl
> REPLACE THIS LINE WITH YOUR PAPER IDENTIFICATION NUMBER (DOUBLE-CLICK HERE TO EDIT) <
Charge Density Study using Low Electrode
Diameter in Epiretinal Prosthesis
Diego Luján Villarreal, Dietmar Schroeder and Wolfgang H. Krautschneider.
Abstract—Reducing electrode size can be advantageous for
Having as a rule of thumb that one should avoid the onset of
stimulating the retina because they produce focal stimulation, i.e.
irreversible Faradaic processes when designing electrical
one active electrode excites a single cell thereby greatly
stimulation systems, this would impose to keep the injected
increasing resolution. The main limitation is, however, the high
charge density at a low level within reversible charge injection
charge density of low electrode area that can cause adverse tissue
reactions. In this study, we analyze the use of rectangular and
linear increase pulse shapes based on charge injection capacity,
A novel strategy has been proposed [3]
that generally
voltage window and threshold current using a single ganglion cell
suggest keeping the pulse width narrower because it confines
model with PEDOT-NaPSS arranged electrode array. We found
the amount of current that can be delivered by a stimulator,
that 100µs linear increase pulse shape delivers a better response
especially if it is battery operated, and provides the minimum
of charge density and electrode potential than rectangular that
charge that occurs when pulse width is of tens of µs.
would avoid irreversible Faradaic reactions.
There is evidence that single linear increase pulse shape at
Index Terms—charge injection capacity, linear increase pulse,
lower pulse durations can deliver lower charge than
low electrode, rectangular pulse shape, retinal implant, voltage
rectangular, linear decrease and sinusoidal pulse shapes [7].
Furthermore, electrolysis of water occurs as a result when
maximum cathodic and anodic potential across the electrodes surpass the "water window" boundary [8]. The water window
is a potential range that is defined by the reduction of water,
ETINAL prosthetic devices have strived to replace the
forming hydrogen gas, in the negative direction, and the
R unctionality of photoreceptors lost because of oxidation of water, forming oxygen, in the positive direction
degenerative diseases such as retinitis pigmentosa (RP) or age-
which may cause corrosion.
related macular degeneration (AMD). These diseases are
Once the electrode potential attains either of these two
incurable by current treatments. Although the ability to create
voltage window boundaries, all further injected charge goes
visual sensations is now well established [1], the quality of
into the irreversible Faradaic processes of water oxidation or
vision elicited by retinal implants is facing further challenges
water reduction [3].
for safely electrode implantation.
PEDOT is a conductive polymer has been generated
Decreasing electrode dimensions will allow focal excitation
considerable attention as a supercapacitor material due to its
of small groups of cells that lead to high resolution patterns of
large electroactive voltage window, high chemical stability
prosthetic-elicited activity and improve visual reception. This
among conductive polymers [9], lower impedance and higher
challenge, however, requires higher charge density that can
charge injection capacity,
QINJ [10].
QINJ is defined as the
cause breakdown of the electrode as well as adverse tissue
amount of charge per unit area that can be delivered through
an electrode without causing water electrolysis.
The electrochemical reactions at the electrode-tissue
The aim of this study is to investigate the use of rectangular
interface, i.e. capacitive double-layer charging, reversible
and linear increase pulse shapes based on charge injection
Faradaic and irreversible Faradaic reactions, carry out charge
capacity, voltage window and threshold current using a single
injection into the neural tissue [3,4,5]. The latter reaction can
ganglion cell model with PEDOT-NaPSS arranged electrode
produce electrolysis of water that leads to localized pH
array. We give some advice on carrying out efficient
changes [6], gas bubble formation that thought to be harmful
stimulation by avoiding damage to the tissue and to the
and physically disturbs the tissue [4, 5] and chemical species
formation that damage the tissue or the electrode [3].
Manuscript received month, day, 2016; revised month, day, 2016; accepted
A. Ganglion Cell Model
month, day, 2016.
Diego Lujan Villarreal, Dietmar Schroeder and Wolfgang H.
Ganglion cell model has a basic mathematical structure for
Krautschneider are with the institute of Nano and Medical Electronics at the
voltage-gating based on Hodgkin and Huxley like equations
Hamburg University of Technology (corresponding author phone: +49 40
[11] and is modelled with an equivalent circuit taken from
42878 3991; fax +49 40 42878 2877
previously published model of repetitive firing of retinal
> REPLACE THIS LINE WITH YOUR PAPER IDENTIFICATION NUMBER (DOUBLE-CLICK HERE TO EDIT) <
ganglion cells [12]. The parameters and equations that
figure 1. In this work we used the identical retinal model as in
describe the dynamics of the ionic channels were kept as in the
The ganglion cell soma was placed inside the retina
ganglion cell layer exactly below the center of active electrode
B. Retinal Model
and was enclosed with the cell membrane.
We used the identical COMSOL model of the retina as seen
In previous published works [16,17,18,19,20] there have
in [13]. The model is shown in figure 1.
been sufficient evidence that monophasic pulse allows the formation of Faradaic reduction reactions. If oxygen is
Polyimide electrode carrier
presence, these reactions may include reduction of oxygen and
CROSS SECTION PLANE
formation of reactive oxygen species associated in tissue
Vitreous medium, VM
Although monophasic is the most efficient pulse for
Retina ganglion layer
stimulation because of the action potential initiation and the
Photoreceptor layer
potential becomes insufficiently positive (using cathodic
Retinal pigment epithelium
pulses) where electrode corrosion may occur, however, it is
not used in continuous pulses where tissue damage is to be
Fig. 1. Retina model at COMSOL simulations. Layer thicknesses not drawn
We used, however, monophasic rectangular and linear
increase shapes with a single anodic pulse for the solely
It consists of seven domains: polyimide carrier of electrodes
intention to reduce the computational time consumed.
PC; vitreous medium VM; retina ganglion cell layer RGC;
We iterated the retinal model for each ED, IEGD and ∆t until
photoreceptor layer PRC; retinal pigment epithelium RPE;
we match the average boundary current density of the cell
ganglion cell soma SG and the electrode array EELE. The
with the extracellular threshold current amplitude obtained in
ganglion cell soma was placed inside the retina ganglion cell
Matlab by applying current from the active electrode.
layer exactly below the center of active electrode and was
We assumed that the irreversible Faradaic reactions, if
enclosed with the cell membrane.
present, will occur such that the charge density surpass the
The material coating the electrode is PEDOT-NaPSS
QINJ limit or the anodic peak potential at the electrode surpass
electrodeposited in gold electrodes with a charge density of 40
the voltage window boundary.
mC/cm2 as seen in [10].
The inter electrode ganglion cell distance, IEGD, are 2, 10,
The electrode array configuration is shown at the cross
100 µm. The electrode diameter, ED, are 2, 10 50, 100 µm.
section plane in figure 1. This arrangement is analogous to
The ∆t are 50 and 100 µs. We briefly analyzed pulse duration
[14,15] however it consists of an active electrode (in red)
of 150 µs and yielded the highest charge density than 50 and
surrounded by eight guards (in blue) in order to stress the
isolation of the active electrode, to confine the stimulus
Out of COMSOL simulations, we also obtained the voltage
current to a small volume around the ganglion cell and to
across the electrodes over time.
minimize electrode cross-talk during stimulation.
E. Charge Density CalculationI
C. Simulation Procedure – Ganglion Cell Model (Matlab)
The charge density was obtained by integrating the current
In this work we used the identical ganglion cell model as in
delivered by the active electrode over time and dividing it by
[12] to calculate in Matlab the extracellular threshold current
the electrode area. It is worth to mention that all eight
density of the ganglion cell by applying monophasic
surrounding electrodes including the active changed their
rectangular and linear increase pulse shapes to the model at
dimensions accordingly.
500 pulses per second, taking into account absolute and
QINJ of gold microelectrode coated with PEDOT-NaPSS
refractory period of an action potential.
follows a linear relationship with charge density used during
We followed the strategy as seen in [3] and we chose two
electropolymerization,
QD, with a constant 0.075
QINJ per
QD
pulse duration, ∆t, of 50 and 100 µs. Pulse widths lower than
(until 300mC/cm2) for 0.001 and 1 mm2 electrode size [10].
150 µs are analogous in [2] to directly stimulate the ganglion
QINJ may also be increased by increasing electrochemical
cell and to elicit solely a single spike with precise temporal
surface area [21].
pattern. The peak current density amplitude was swept with a
As there is no evidence about
QINJ in PEDOT-NaPSS low
resolution of 1 µA/cm2 until it was found the threshold current
electrode area, we used the limit of 0.35 mC/cm2 for gas-free
density that fires a train of action potential.
and erosion-free operation [4] and 1mC/cm2 for neural
The result of extracellular peak current amplitude are 330
and 120 µA/cm2 for rectangular pulse shape and 340 and 160
F. Voltage Window Boundary
µA/cm2 for linear increase at 50 and 100 µs, respectively.
PEDOT voltage window extends beyond conductive
D. Simulation Procedure – Retinal 3D Model (COMSOL)
materials, such as MnO2, from 1.5 V [23] up to 1.7 V [9].
The retinal modelling was built in COMSOL and shown in
As there is no evidence about voltage window on PEDOT-
> REPLACE THIS LINE WITH YOUR PAPER IDENTIFICATION NUMBER (DOUBLE-CLICK HERE TO EDIT) <
NaPSS low electrode area, we used the limit of 1.7 V.
B. Charge Density
It is evident that the charge density, figures 2 to 4, decreases
using linear increase pulse shape.
A. Charge Density Results
Figures 2 to 4 show the comparison of threshold charge
density, left y-axis, and the threshold current for ganglion cell activation, right y-axis, for monophasic rectangular and linear increase pulse shapes.
Each plot shows the results for a specific IEGD. On top of
each plot the forbidden region of gas formation and neural damage are shown with a red dashed-line.
Table I lists the minimum electrode diameter [µm] that can
be used with their corresponding limit.
The green boxes indicate the suitability to use the minimum
electrode diameter tested of 2 µm.
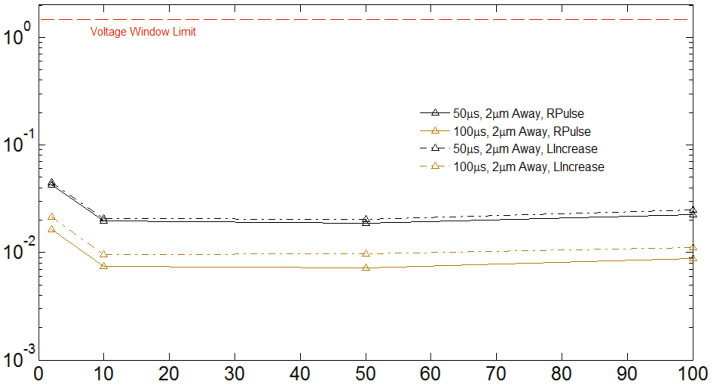
Fig. 2. Threshold charge density and threshold current for 2 µm inter
electrode-ganglion cell distance.
HARGE DENSITY LIMITS FOR RECTANGULAR/LINEAR INCREASE PULSE
Using 50 µs pulse duration, charge density is reduced up to
0.35 mC/cm2 limit
40±2.4% in average; for 100 µs, it is decreased 30±1.3%. This
means a promising technique to avoid irreversible Faradaic
The rectangular pulse shape has its own attributes meaning
that using ED of 2 µm, 50 or 100 µs pulse duration, and IEGD
lower than 10 µm is safe within the limits. This technique
For a successful implant of retinal device, only the lower
works with linear increase pulse shape as well. Doing so, it
safe charge density of 0.35mC/cm2 should be employed.
provides a method to send a more natural signal to the brain and to generate meaningful percepts.
B. Voltage across Electrodes Results
Figure 4 show the constraint of charge density at both limits
Figures 5 and 6 illustrate the comparison of voltage across
mainly when IEGD is greater than 10 µm.
the electrodes. Each plot shows the results for a specific IEGD.
On top of each plot the voltage window boundary is shown
with a red dashed-line.
Table II lists the minimum electrode diameter [µm] that can
be used for the corresponding limit.
VOLTAGE LIMITS FOR PULSE SHAPES
Fig. 3. Threshold charge density and threshold current for 10 µm inter
The green boxes indicate the suitability to use the minimum
electrode-ganglion cell distance.
electrode diameter tested of 2 µm.
For achieving a better response in continuous pulses,
charge-imbalanced biphasic waveform provides a method to
A. Threshold Current
reduce the irreversible charge density as reactions occurring in one phase (cathodic or anodic) are reverse in the following [3].
Threshold currents, figures 2 to 4, were found to increase
Furthermore, it was demonstrated that this waveform allows
with time after surgery, most likely due to the lifting off of the
greater cathodic charge densities than monophasic prior to the
electrode array from the retinal surface [24].
onset of tissue damage [26].
Threshold variations with respect to IEGD are consistent with
previous experimental work of epiretinal device implanted in
C. Voltage across Electrodes
It is also evident that using 100 µs pulse duration, more than
Because linear increase shape injects less charge than
one third but less than half of electrode voltage, fig. 5 and 6, is
rectangular for a given pulse duration, the former necessitates
reduced than 50 µs for both pulse shapes. This technique
a higher amplitude to reach threshold.
would avoid irreversible Faradaic reactions.
> REPLACE THIS LINE WITH YOUR PAPER IDENTIFICATION NUMBER (DOUBLE-CLICK HERE TO EDIT) <
For attaining a better response in successive pulses, the
charge-imbalanced waveform has advantages in avoiding corrosion by decreasing the maximum positive potential since the anodic phase is no longer constrained to be equal to the cathodic phase as charge-balanced pulse, thus the electrode potential reaches less positive values [3,26].
We found that 100µs linear increase pulse shape delivers a
better response of charge density and electrode potential than rectangular that would avoid irreversible Faradaic reactions.
Fig. 4. Threshold charge density and threshold current for 100 µm inter
Furthermore, for a given IEGD with charge density limit of
electrode-ganglion cell distance.
0.35mC/cm2 and voltage window limit of 1.7V, our model
Considering figure 5 and 6, we learn that it is safe to work
i) 0<IEGD<10 µm: a) reduce electrode diameter to 2 µm; b)
work with either 50 or 100 µs low pulse duration; c) use either
D of 2 µ m, low pulse durations lower than 150 µ s, IEGD
lower than 10 µm with either rectangular or linear increase
rectangular or linear increase pulse shapes.
ii) 10<IEGD<100 µm (for rectangular pulse): a) reduce
electrode diameter to 14 µm only with 100 µs pulse duration. For 50 µs, electrode diameter should be 18 µm.
iii) 10<IEGD<100 µm (for linear increase pulse): a) reduce
electrode diameter to 10 µm with either 50 or 100 µs pulse duration.
If pulse trains are needed, the charge-imbalanced waveform
has added advantages in avoiding corrosion and reducing irreversible charge densities that leads to either electrode or tissue damage [3,26].
Additional experimental testing of small electrodes is still
required to verify our results. Moreover, further simulations of heat dissipation should be performed to verify the use of
Fig. 5. Voltage results for 2 µm inter electrode-ganglion cell distance for
1000+ electrode array for epi- or subretinal implants.
rectangular and linear increase pulse shapes.
Figure 6 show that the constraint of voltage window limit
[1] Cai, Qiushi, et al. Response variability to high rates of electric
originates mainly when IEGD is greater than 10 µm.
stimulation in retinal ganglion cells. J Neurophysiology, 2011.
[2] Fried S., et al. A Method for Generating Precise Temporal Patterns of
Retinal Spiking Using Prosthetic Stimulation. J Neurophysiol 95: 970–978, 2006.
[3] D. R. Merrill, M. Bikson, and J. G. Jefferys, "Electrical stimulation of
excitable tissue: Design of efficacious and safe protocols,"J. Neurosci. Methods , vol. 141, no. 2, pp. 171–198, Feb. 15, 2005.
[4] S. B. Brummer and M. J. Turner, "Electrochemical considerations for
safe electrical stimulation of the nervous system with platinum electrodes," IEEE Trans. Biomed. Eng., vol. 24, no. 1, pp. 59–63, Jan. 1977.
[5] S. F. Cogan, "Neural stimulation and recording electrodes," Annu. Rev.
Biomed. Eng., vol. 10, pp. 275–309, 2008.
[6] C. Q. Huang, P. M. Carter, and R. K. Shepherd, "Stimulus induced pH
changes in cochlear implants: An in vitro and in vivo study," Ann. Biomed. Eng., vol. 29, no. 9, pp. 791–802, Sep. 2001.
[7] Mario A. Meza-Cuevas, Dietmar Schroeder and Wolfgang H.
Fig. 6. Voltage results for 10 and 100 µm inter electrode-ganglion cell
Krautschneider. Neuromuscular Electrical Stimulation Using Different
distance for rectangular and linear increase pulse shapes.
Waveforms - Properties comparison by applying single pulses. 5th
International Conference on BioMedical Engineering and Informatics,
It should be noted that in fact irreversible processes might
occur at potentials within the voltage window, such as
[8] Ronald T. Leung, Mohit N. Shivdasani, David A. X. Nayagam, Robert
K. Shepherd. In Vivo and In Vitro Comparison of the Charge Injection
irreversible oxygen reduction, as opposed to the opinion in
Capacity of Platinum Macroelectrodes. IEEE Transactions on
many studies that states reversible charge storage capacity, or
Biomedical Engineering, Vol. 62, No. 3, March 2015.
[9] Jonathon Duay, Eleanor Gillette, Ran Liu, Sang Bok Lee. Highly
INJ, can be applied without the electrode potential exceeds
flexible pseudocapacitor based on freestanding heterogeneous
the voltage window during pulsing [3]. Therefore, QINJ and
MnO2/conductive polymer nanowire arrays. Phys. Chem. Chem. Phys.,
voltage window limits need to be analyzed separately.
2012, 14, 3329–3337.
> REPLACE THIS LINE WITH YOUR PAPER IDENTIFICATION NUMBER (DOUBLE-CLICK HERE TO EDIT) <
[10] Starbird, R. et al. Electrochemical properties of PEDOT-NaPSS
He has worked with the Cellular Therapy
galvanostatically deposited from an aqueous micellar media for invasive
Department in the School of Medicine at
electrodes, IEEE BMEICON, 2012.
[11] Hodgkin A., et al. A quantitative description of membrane current and
ITESM, Monterrey, México, and he is
its application to conduction and excitation in nerve. J. Physiol, 1952.
now pursuing his PhD in the Institute of
[12] Fohlmeister J., et al. Modeling the repetitive firing of retinal ganglion
Nano and Medical Electronics at the
cells. Brain Research, 1989.
Hamburg University of Technology,
[13] Lujan Villarreal D., et al. "Feasibility Study of a 1000+ Electrode Array
in Epiretinal Prosthesis." (Forthcoming, 2016) 8th International
Hamburg, Germany.
Conference on Bioinformatics and Biomedical Technology. Barcelona,
His main research interests focus on
Spain. June 10-12, 2016.
theoretical models of neurostimulation, optimal energy-saving
[14] Lovell N., et al. Current distribution during parallel stimulation:
algorithms and epi- and sub retinal stimulation.
Implications for an epiretinal neuroprosthesis. IEEE Eng Med Biol Soc,
[15] Dommel N., et al. A CMOS retinal neurostimulator capable of focussed,
simultaneous stimulation J Neural Eng, vol. 6, pp. 035006, 2009.
[16] Halliwell B. Reactive oxygen species and the central nervous system. J
Neurochem 1992;59(5):1609–23.
Schroeder
[17] Imlay JA. Pathways of oxidative damage. Annu Rev Microbiol
2003;57:395–418.
received the Dr. Ing. degree in electrical
[18] Bergamini CM, Gambetti S, Dondi A, Cervellati C. Oxygen, reactive
oxygen species and tissue damage. Curr Pharm Des 2004;10(14):1611–
Universität Braunschweig, Braunschweig,
Germany, in 1984.
[19] Stohs SJ. The role of free radicals in toxicity and disease. J Basic Clin
Physiol Pharmacol 1995;6(3–4):205–28.
He joined the Hamburg University of
[20] Hemnani T, Parihar MS. Reactive oxygen species and oxidative DNA
Technology, Hamburg, Germany, in
damage. Indian J Physiol Pharmacol 1998;42(4):440–52.
1983, where he has been a Lecturer with
[21] Mario A. Meza-Cuevas. Stimulation of Neurons by Electrical Means.
Semiconductor Electronics since 1994.
Logos Verlag Berlin GmbH. 2015.
[22] Warren E. Finn, et al. Handbook of Neuroprosthetic Methods. CRC
Press, Dec 16, 2002.
[23] Boretius, T., M. Schuettler, and T. Stieglitz. On the Stability of PEDOT
as Coating Material for Active Neural Implants. 15th Annual
Conference of the International Functional Electrical Stimulation Society, 2010.
Wolfgang H. Krautschneider (M'85)
[24] Chader G., et al. Artificial vision: needs, functioning, and testing of
retinal electronic prosthesis. Progress Brain Research, Vol. 175, 2009.
Habilitation degrees from the Berlin
[25] Jensen R., et al. Thresholds for Activation of Rabbit Retinal Ganglion
Cells with Relatively Large, Extracellular Microelectrodes. IOVS, 2005.
[26] Scheiner A, Mortimer JT. Imbalanced biphasic electrical stimulation:
muscle tissue damage. Ann Biomed Eng 1990.
Laboratories, IBM, Yorktown Heights,
Diego Lujan Villarreal was born in Monterrey, Nuevo León,
NY, USA, the Siemens Research Center,
México in 1983. He received the B.S. degree in Mechatronics
Munich, Germany, and the DRAM
from the Monterrey Institute of Technology and Higher
Project of IBM and Siemens, Essex Junction, VT, USA.
Education (ITESM) in 2006 and the M.S. degree in
He is currently the Head of the Institute of Nano and Medical
Microelectronics and Microsystems from the Hamburg
Electronics at the Hamburg University of Technology,
University of Technology in 2010.
Hamburg, Germany.
Source: http://www.ictopen.nl/binaries/content/assets/bestanden/ict-open-2016/proceedings/villarreal-diego-lujan.pdf
sb9103.qxd 12/16/1999 9:19 AM Page 48 Combinatorial gene regulation by eukaryotic transcription factorsLin Chen Recent structure determinations of high order complexes of eukaryotic transcription factors bound to DNA have revealedthat residues from their DNA-binding domains are involved in MCM1 MADS box dimer protein–protein interactions between distinct factors.
INCIDENCE OF PUBIC OSTEOMYELITIS AFTER BLADDER NECK SUSPENSION USING BONE ANCHORS ROGER P. GOLDBERG, MARIE BLANCHE TCHETGEN, PETER K. SAND, SUMANA KODURI, RAYMOND RACKLEY, RODNEY APPELL, AND PATRICK J. CULLIGAN Objectives. To determine the incidence of pubic osteomyelitis after bladder neck suspension using supra-pubic bone anchors.Methods. The target population consisted of 290 consecutive women who underwent bladder necksuspension using suprapubic bone anchors between June 1994 and November 1999 at two referral centers.A structured telephone questionnaire was designed to elicit any history of clinical symptoms suspicious forpubic osteomyelitis. Positive responses were followed up by a detailed review of the medical records.Nonresponders were evaluated by chart review, with negative cases included only if the documentedfollow-up reached 1 year.Results. The sample consisted of 225 women, representing 77.6% of the study population, with a mean ageof 69.7 years (range 40 to 88) and a mean follow-up of 31.8 months (range 13.4 to 42.2). Of the 225women, 179 (80%) completed the telephone survey; 46 patients (20%) were evaluated by long-term chartreview. Three patients (1.3%) reported positive responses to the screening questionnaire and were con-firmed to have developed pubic osteomyelitis. Each had undergone exploratory laparotomy, anchor re-moval, bony debridement, and prolonged parenteral antibiosis. The most common noninfectious complaintswere irritative voiding symptoms and pubic or groin pain responding to "conservative" therapy (3.5%),including 1 case of osteitis pubis. One subject underwent repeated operation because of erosion of the slingsutures into the bladder.Conclusions. The estimated incidence of osteomyelitis after bone-anchored bladder neck suspension was1.3%. Although postoperative osteomyelitis is rare, each case incurs substantial morbidity and a compli-cated postoperative course. UROLOGY 63: 704–708, 2004. © 2004 Elsevier Inc.

