Levitra enthält Vardenafil, das eine kürzere Wirkdauer als Tadalafil hat, dafür aber schnell einsetzt. Männer, die diskret bestellen möchten, suchen häufig nach levitra kaufen ohne rezept. Dabei spielt die rechtliche Lage in der Schweiz eine wichtige Rolle.
Mg_handbook-english-and-french-final.indd
A Guide to the Diagnosis & Management of Myasthenia Gravis
Joël Oger, MD, FRCPC, FAA.
Dr. John Newsom-Davis 1932-2007
Expert reviews:
J. Newsom-Davis CBE, MA, FRCP, FRS,
Design & illustration:
L.Waters, MFA, MScBMC, Waters Biomedical
Acknowledgements:
I thank Talecris Biotherapeutics for making this booklet possible through an unrestricted
educational grant, and Kim Fisher, PhD, for her support. I would also like to thank
Lori Waters for the illustration and design.
A Guide to the Diagnosis & Management of Myasthenia Gravis
Joël Oger, MD, FRCPC, FAA.
Dr. John Newsom-Davis 1932-2007
Expert reviews:
J. Newsom-Davis CBE, MA, FRCP, FRS,
Design & illustration:
L.Waters, MFA, MScBMC, Waters Biomedical
Acknowledgements:
I thank Talecris Biotherapeutics for making this booklet possible through an unrestricted
educational grant, and Kim Fisher, PhD, for her support. I would also like to thank
Lori Waters for the illustration and design.
To order your copy, please visit: < www.myastheniagravisbooklet.com >or send us an email. Dr. Joël Oger: <
[email protected]>
Table of Contents
Introduction
Section 1: Autoimmune Myasthenia Gravis
Section 2: Diagnostic Tests
Electromyography (Repetitive Stimulation EMG)
Electromyography
Section 3: Additional Tests
Staging of Disease - Osserman Classifi cation of MG
Quantitative Myasthenia Gravis Examination (QMG)
Section 4: Treatment
Acetylcholinesterase
Immunosuppressive
Table of Contents (continued)
Section 5: Special Considerations
Myasthenia Gravis during Pregnancy
Myasthenia Gravis in the Elderly
Section 6: MG Variants
Lambert Eaton Myasthenic Syndrome (LEMS)
Section 7: Appendices
A1: List of Contra-indicated Medications in MG
A2: Quantitative Myasthenia Gravis Exam (QMG)
A5: Author Contact Information
Introduction to the reader
During my many years of following Myasthenia Gravis (MG) patients, I have noticed that physicians adhere to relatively similar therapeutic plans. Most helpful to MG patients, however, is the practitioner's att ention to details that include a clear and exhaustive plan for drug titration. This detailed approach will make the patient confi dent that he or she has found the doctor in whom he or she can put his or her confi dence.
This booklet has been writt en with neurology residents in mind as an easy reference document that they can access when they have to admit their fi rst MG patient. It will also be useful to neurologists in general practice who need a refresher because one does not see a new MG patient oft en. It could also help other health care professionals when the time comes to counsel patients. I have oft en suggested to my patients to off er this type of booklet to their family doctors as soon as they have been diagnosed.
Section 1: Autoimmune Myasthenia Gravis (MG) — Classifi cation
Acquired or Autoimmune Myasthenia Gravis (MG)
• Bulbar Predominant MG
• Generalized MG
Classifi cation of Autoimmune Myasthenia Gravis (MG) based on Antibodies
• Acetylcholine Receptor Antibody positive MG
• Anti–MuSK (muscle specifi c kinase)Antibody Positive MG
• Antibody Negative Autoimmune MG
Forms of Autoimmune Myasthenia Gravis based on age at onset •
Autoimmune Myasthenia Gravis (MG): Epidemiology and Pathophysiology
Myasthenia Gravis is a rare disease with a prevalence of 20/100,000. Most neurologists would not be expected to see more than 5 or 6 in their whole career unless theyspecialize in neuromuscular disorders or neuro-immunology. Similar to what is seenin other autoimmune disorders, there is evidence of a genetic predisposition andcertain human leukocyte antigen (HLA) associations. There is no racial or ethnicpredominance.
Two age groups have been identifi ed as being preferentially aff ected: young, early onset (< 45 years of age; a group where women tend to predominate) and elderly, late onset (> 60 year of age; a group where men tend to slightly predominate).
As time passes, the disease becomes more common and is more frequently recognized among those over the age of 80, probably evidence of successful aging of the Western population and availability of testing.
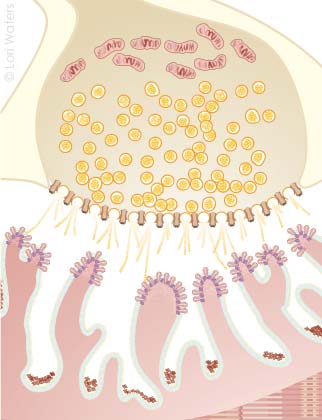

Figure 1. Physiology of the normal neuromuscular junction (NMJ)
Motor neuron axonMitochondria
Synaptic vesicles
Calcium ion channels
Synaptic junction
Acetylcholine receptors
When the action potential reaches the NMJ, Ca++ fl ows into (1) the voltage-gated channels releasing acetylcho-
line (ACh) from the presynaptic vesicles (2). ACh binds to the receptor at the top of the post junctional folds of the
muscle membrane (3). Binding of ACh to the receptor results in opening of the channel pore (insert) permitting
Na+ infl ux in the muscle and initiating muscle contraction. Excess ACh is destroyed by acetylcholinesterase (4).
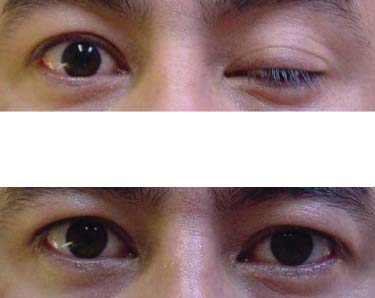
Autoimmune Myasthenia Gravis: Ocular MG — Signs and Symptoms (cont'd)
1 Left ptosis (Ocular MG)
2 Aft er icepack fi ve minutes.
(see Ice cube test —page 23).
Additional Tests: Staging of Disease
Osserman Classifi cation of Myasthenia Gravis (modifi ed from MGFA)
Class I – pure ocular
• Any ocular weakness, may have weakness of eye closure;
• Strength of all other muscles is normal.
Class II – ocular plus other defi cits
• IIa Mild weakness aff ecting limbs±bulbar muscles;
• IIb Predominantly aff ecting limbs ±oropharyngeal.
Class III – moderate generalized
• IIIa Moderate weakness Predominantly aff ecting limb, axial muscles, or both;
• IIIb Moderate generalized weakness predominantly aff ecting oropharyngeal,
respiratory muscles, or both.
Class IV – severe generalized
• IVa Severe weakness aff ecting limbs;
• IVb Severe weakness aff ecting limbs. Predominantly aff ects bulbar muscles.
Class V
• Ventilator dependent except when used during routine postoperative management.
Table 2 - Acetylcholinesterase Inhibitors
Administration Route /
Dosage Form
Dose / Regimen
Therapeutic Eff ect
oral (tablet; Mestinon®)
Initial: 60-120 mg q
3-4 hrs during wak-ing hours
oral (extended-release
tablet; Mestinon®-SR)**
times/day (12 hrs between doses)**
oral (syrup; not commer-
cially available can be
made by a compound-ing pharmacist)
IV (injection USP;
** poor bioavailability so should be used only at bedtime to relieve early morning symptoms
Administration Route /
Dosage Form
Dose / Regimen
Therapeutic Eff ect
oral (tablet; Prostigmin®)
Initial: 15 mg q 3-4
hrs. Maintenance: 150 mg/day (24-hr period)
Administration Route /
Dosage Form
Dose / Regimen
Therapeutic Eff ect
IM, SC (injection USP;
0.5-2.5 mg every
1-3 hours (up to 10
Administration Route /
Dosage Form
Dose / Regimen
Therapeutic Eff ect
IV (injection USP;
0.5-2 mg administered slowly,
every 1-3 hours (up to 10
mg/24 hrs maximum)
Treatment: Corticosteroid Agents (cont'd)
The following treatment protocol for prednisone has been eff ective and safe:
• Starting dose of 10 mg every other day.
• Increase dose by 5 to 10 mg every 4th day to reach 1 mg/kg/day every other day.
• Continue at this dose until patient reaches remission.
• Symptoms rarely worsen with this scheme. If worsening occurs, then treat with
plasma exchange or IVIg.
• When the patient reaches remission, reduce and discontinue Mestinon®.
An inability to discontinue Mestinon® indicates that patient is not well controlled.
• When remission (or a minimal symptomatic state) has been achieved, prednisone
can be reduced progressively by 10 mg every 2 weeks to a dose of 50 mg every other
day. Then by 5 mg every month until 25 mg e.o.d.
• Prednisone reduction is more easily achieved if an immunosuppressive drug is
added.
During the reduction in prednisone dosage:
• The patient should be observed closely for re-appearance of signs and symptoms.
• If signs re-appear, it is necessary to increase the prednisone back to the dose at which
the patient was minimally symptomatic.
Treatment: Immunosuppressive Therapies — Azathioprine & Cyclosporin A
Azathioprine (Imuran®) (Aza.)
• Recommended as fi rst choice steroid-sparing medication;
• Aza. may take 12 months to be eff ective, but cyclosporine acts faster in a matt er of a
few weeks. It is still the only immunosuppressive agent shown to improve MG in an
RCT (Palace et al).
• Generally well tolerated; rarely an acute idiosyncratic reaction occurs on initiation of
therapy with fever and hepatitis. Treatment should be stopped immediately.
• Aza. has not been shown to be harmful to the fetus. Numerous uncomplicated
pregnancies have occurred in transplant patients receiving Aza.
• Dosage with Aza. should rapidly reach 3mg/kg/day in 3 divided doses (gastric
tolerance permitt ing) and the dose should be adjusted to the monthly blood work
(CBC, WBC, Diff , platelets and ALP). See sliding scale on page 77.
• The administration of Aza. can reduce bone marrow maturation and lead to liver
dysfunction with a micronodular cirrhosis.
Treatment: Intravenous Immunoglobulins (IVIg)
Mechanism of Action
• In Myasthenia Gravis autoantibodies are pathogenic and are of the IgG class.
• IVIg reduces the amount of antibodies bound at the neuromuscular junction by
increasing the total concentration of IgG circulating in serum and extra-cellular fl uids.
IVIg also modulates regulatory cells by binding to Fc receptors & anti-idiotypes.
Indication
• IVIg is useful in myasthenic crisis, in preparing MG patients for surgery, and aiding
diagnosis of seronegative MG.
• Available in areas without plasmapheresis centres. Its single draw back compared to
use of plasma exchange is that it may act slightly more slowly.
• It is important to stress that IVIg does not stimulate the immune response so that it
can be used chronically in very resistant cases.
• IVIg is expensive with a cycle of 2mg/kg/day costing in excess of $10,000.
Side Eff ects
• Allergic reactions, headache, occasional lymphocytes meningitis, clott ing problems;
• Some premedicate patients with anti-histamine (Benadryl®).
Table 4: Intravenous Immunoglobulin (IVIg) versus Plasma Exchange (Plex)
Intravenous Immunoglobulin (IVIg)
Plasma Exchange (Plex)
Mechanism of Action
Downregulates production of anti-AChR Removes anti-AChR antibodies from extra-antibodies; modulates T-cell function
Exacerbations of MG; myasthenic crisis;
Exacerbations of MG; myasthenic crisis;
preparation for surgery; long term use
preparation for surgery
2 g/kg divided over 2-5 days
3 to 5 exchanges, daily or every second day
Onset of Action
Duration of Eff ect
1-2 months (IVIg = Plex)
1-2 months (IVIg = Plex)
Side Eff ects
Platelet aggregation problems, Allergic
BP variations, venous access problems,
reactions, Lymphocytic meningitis (rare)
duration of procedure= 3-5 hours,
$5,120 + indirect costs / sitting
Widely available - infused in outpatient
Requires special equipment & expertise;
available in very few tertiary care centres
MG Variants — Lambert Eaton Myasthenic Syndrome (LEMS) (cont'd)
• Presence of voltage-gated calcium channel antibodies in >90% of patients.
• Surgical removal of the primary neoplasm may greatly improve LEMS symptoms.
• Symptomatic treatment includes 3, 4-diaminopyridine (10-20mg four times/day-risk of seizure with higher doses.), quinidine, or 4-aminopyridine. Mestinon® by itself is not eff ective but can work synergistically with 3, 4-diaminopyridine.
• Immune directed treatment can be added when tumour removal is insuffi
it will be the sole treatment for the autoimmune form: dosage of Azathioprine is same as in MG (see page 53).
• IVIg (2 g/kg, divided over 2-5 days) as an adjunct to immunosuppressive therapy.
• Plasmapheresis would only be used as induction.
Repetitive Stimulation in LEMS
Appendix 1: Medications Contra-indicated in Myasthenia Gravis
1. CURARE Derivatives but also
• Propranolol, Oxprenolol, Practolol &
Methoxyfl urane (Penthrane®)
• other beta blockers
Accupril ® & other ACE inhibitors
Verapamil and Calcium Channel
(mostly aminoglycosides)
• Neomycin, Streptomycin, Gentamicin
• Kanamycin, Tobramycin, Amikacin
4. CNS ACTIVE DRUGS
• Polymyxin B, Colistins
• Chlorpromazine and Promazine
• Tetracyclines,
• Erythromycin
• Diphenylhydantoin &
• Azithromycin
• Trihexyphenidyl (Artane®)
• Trimethaphan (antitussif
in many cough preps.)
• Ketek (Telithromycin)
• D-Penicillamine
3. CARDIOVASCULAR DRUGS
in bold are
dangerous
• Procaine, Procainamide, Lidocaine
• Labetalol chlorhydrate (Trandate®)
Appendix 4: Further Reading
Evaluation of diagnostic tests: A systematic review of diagnostic studies in MG
Michael Benatar Neuromuscular disorders 2006;16:459-467.
Seronegative generalized myasthenia gravis: Clinical features, antibodies and their
targets A. Vincent, J. Bowen, J. Newsom-Davis and J. McConville Lancet 2003
The single report of RCT in immunosuppression of MG: J. Palace, J. Newsom-Davis,
B Leaky and the MG study group RCT of prednisolone alone or with
azathioprine in Myasthenia Gravis Neurology 1998:50; 1778-1783
Myasthenia Gravis Foundation of America's: Recommendations for Clinical Research
Standards A. Jaretski III, R. Barohn et al. Ann Thor Surg 2000, 70:327-334
Sharshar et.al. J.Neurol.2000; 247: 286-290.
Congenital Myasthenic syndromes: progress over the last decade A. Engel, K. Ohno
and SM Sine: Muscle nerve 2003 (27): 4-25
Medications to avoid in MG: Z. Argov and I Wirguin: Drugs and the neuromuscular
junction in Handbook of Myasthenia Gravis and Myasthenic Syndromes
pp 295-315 R.P.Lisak Ed 1 vol.Marcel Dekker New York, Basel Hong Kong
1994) Look for regular updates under Z. Argov
Author Contact Information
We welcome your comments. If you fi nd errors, or if you have questions, commentsor suggestions, do not hesitate to write to me:
Dr Joël Oger
Neurology
Room S-159
UBC Hospital
2211 Wesbrook Mall
Vancouver BC V6T 2B5
CANADA
email: < [email protected] >
Source: http://www.myastheniagravisbooklet.com/Myasthenia_Gravis_Handbook-Preview.pdf
2ND ANNUAL FLORIDA RESIDENCY CONFERENCE May 9-10, 2013 University of Florida College of Pharmacy Gainesville, Florida Sponsored by the FSHP Research and Education Foundation 2ND ANNUAL FLORIDA RESIDENCY CONFERENCE Welcome to the Second Annual Florida Residency Conference. We are grateful to all in attendance and we are looking forward to another great conference full of high quality resident presentations, as well as some quality time with friends and colleagues. The Florida Residency Conference Steering Committee has been busy looking ahead to not only this conference but the future of the FRC.
Emergency Planning for the Important Safety Information For Your Community and Annual Special Needs Survey Please read the entire brochure or have someone translate it for you. Discuss this information with members of your family, and then keep the brochure in a convenient place for future use. ESTA INFORMACIÓN ES IMPORTANTE



